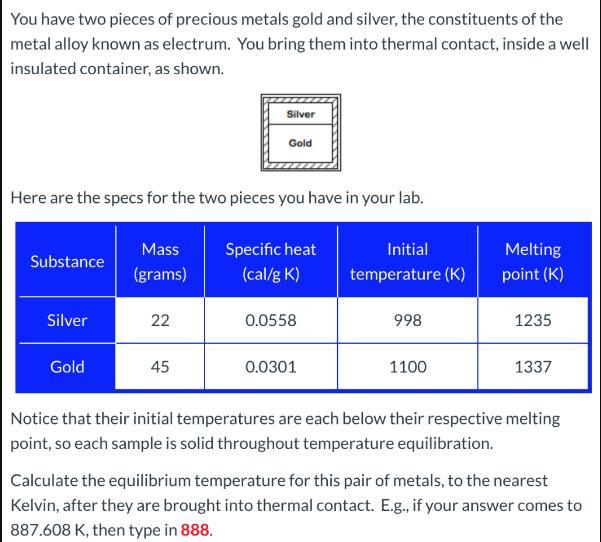
You have two pieces of precious metals gold and silver, the constituents of the metal alloy...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
You have two pieces of precious metals gold and silver, the constituents of the metal alloy known as electrum. You bring them into thermal contact, inside a well insulated container, as shown. Here are the specs for the two pieces you have in your lab. Substance Silver Gold Mass (grams) 22 Silver Gold 45 Specific heat (cal/g K) 0.0558 0.0301 Initial temperature (K) 998 1100 Melting point (K) 1235 1337 Notice that their initial temperatures are each below their respective melting point, so each sample is solid throughout temperature equilibration. Calculate the equilibrium temperature for this pair of metals, to the nearest Kelvin, after they are brought into thermal contact. E.g., if your answer comes to 887.608 K, then type in 888. You have two pieces of precious metals gold and silver, the constituents of the metal alloy known as electrum. You bring them into thermal contact, inside a well insulated container, as shown. Here are the specs for the two pieces you have in your lab. Substance Silver Gold Mass (grams) 22 Silver Gold 45 Specific heat (cal/g K) 0.0558 0.0301 Initial temperature (K) 998 1100 Melting point (K) 1235 1337 Notice that their initial temperatures are each below their respective melting point, so each sample is solid throughout temperature equilibration. Calculate the equilibrium temperature for this pair of metals, to the nearest Kelvin, after they are brought into thermal contact. E.g., if your answer comes to 887.608 K, then type in 888.
Expert Answer:
Answer rating: 100% (QA)
The equilibrium temperature can be found using the following equation Tequilibr... View the full answer

Related Book For 

Posted Date:
Students also viewed these accounting questions
-
Planning is one of the most important management functions in any business. A front office managers first step in planning should involve determine the departments goals. Planning also includes...
-
A sample containing an alkali sulfate is dried, weighed and dissolved in dilute HCl. Barium chloride solution is added in excess to precipitate barium sulfate, and the precipitate is digested in the...
-
According to the 1980 revisions to the Copyright Act of 1976,_____. a. the owner of a copyrighted program is allowed to make multiplecopies or adaptations for use on other computers b. copyright...
-
The Matthew Company uses a normal job-costing system at its Minneapolis plant. The plant has a machining department and an assembly department. Its job-costing system has two direct-cost categories...
-
Verify that the given function is a solution of the differential equation that follows it. u(t) = Ce + Cte; u"(t) - 2u' (t) + u(t) = 0
-
Allan and Koraev both owned condominiums in the same building. Koraevs unit was directly above Allans. While Allan lived in her own unit, Koraev leased his. The leasing of Koraevs unit was managed by...
-
Musixscore.com is an online service providing sheet music to customers. On the browse music Web page, customers select a genre of music from a drop-down list. The Web page uses Ajax to obtain a list...
-
1. (10) A $10 000 bond was issued on January 1st 2015 with a coupon rate of 9.7% and a redemption date of January 1st 2025. What is the purchase price of the bond on January 1st, 2022 when the yield...
-
A company determined that the budgeted cost of producing a product is $ 30 per unit. On June 1, there were 85000 units on hand, the sales department budgeted sales of 360000 units in June, and the...
-
Group Genesis is considering upgrading its computer servers. Two mutually exclusive plans are proposed by the IT consulting firm and their respective estimated net cash flows are listed below. Assume...
-
Please explain the cash collection procedures in accounting information system.
-
Lowell Industries just paid a dividend of D0 = $2.50. Analysts expect the company's dividend to grow by 20% each year for the first two years, and at a constant rate of 5% in Year 3 and thereafter....
-
A 3.5 oz serving of green beans is to be served to each of 130 people at a dinner. How many bags of frozen green beans must you order if each bag contains 5 lbs? How much will it cost if one bag cost...
-
In his meeting with Chairman Xi Jinping in 2021, President Trump hoped to accomplish a 12% revaluation from the current rate of CNY6.74486/$. The exchange rate President Trump is hoping for is CNY...
-
The use of Artificial Intelligence (AI) in Supply Chain management, demand planning, and forecasting: Provide a short history of AI. How do you envision the use of AI in the supply chain in the...
-
Review Exhibit 11.4. Analyze each product on the graph according to the characteristics that influence the rate of adoption. For example, what can you conclude from the data about the relative...
-
Use bond energies to estimate H for the reaction H2(g) + O2(g) H2O2(g)
-
In the following four models, C atoms are black, H atoms are light blue, O atoms are red, and N atoms are dark blue: a. Write the molecular formula of each molecule. b. Write the condensed structural...
-
Sodium-23 is the only stable isotope of sodium. Predict how sodium-20 will decay and how sodium-26 will decay.
-
Enter up a columnar purchases day book with columns for the various expenses for J. Still for the month from the following information on credit items. 2016 January f 1 Bought goods from H. Graham...
-
A Enter up a columnar purchases day book with columns for the various expenses for F. Graham for the month from the following information on credit items. 2016 June Bought goods from J. Syme 4 Bought...
-
Enter up the relevant accounts in the purchases and general ledgers from the columnar purchases day book you completed for Review Question 20.4A. Data From Review Question 20.4A 20.4A Enter up a...

Study smarter with the SolutionInn App


