You pour out 260 g of tea, (specific heat c = 1.00ca. g K' same as...
Fantastic news! We've Found the answer you've been seeking!
Question:
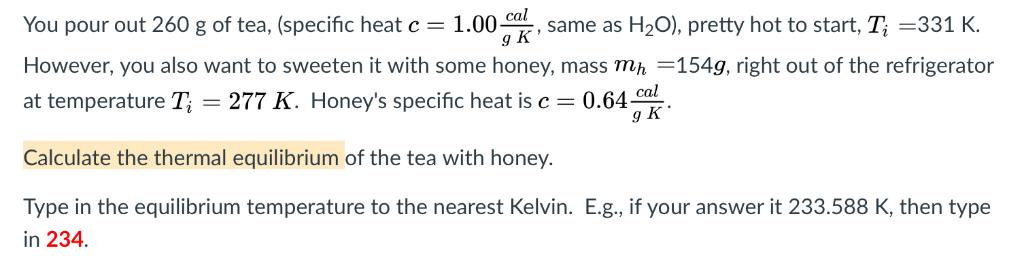
Transcribed Image Text:
You pour out 260 g of tea, (specific heat c = 1.00ca. g K' same as H20), pretty hot to start, T; =331 K. However, you also want to sweeten it with some honey, mass mh =154g, right out of the refrigerator at temperature T; = 277 K. Honey's specific heat is c = 0.64 cal Calculate the thermal equilibrium of the tea with honey. Type in the equilibrium temperature to the nearest Kelvin. E.g., if your answer it 233.588 K, then type in 234. You pour out 260 g of tea, (specific heat c = 1.00ca. g K' same as H20), pretty hot to start, T; =331 K. However, you also want to sweeten it with some honey, mass mh =154g, right out of the refrigerator at temperature T; = 277 K. Honey's specific heat is c = 0.64 cal Calculate the thermal equilibrium of the tea with honey. Type in the equilibrium temperature to the nearest Kelvin. E.g., if your answer it 233.588 K, then type in 234.
Expert Answer:
Answer rating: 100% (QA)
Using Energy conservation Net Energy absorbedreleased by masses 0 Q1 Q2 0 e... View the full answer

Related Book For 

Posted Date:
Students also viewed these accounting questions
-
A Carrot refrigerator is operated between two heat reservoirs at temperatures of 320 K and 270 K. (a) If in each cycle the refrigerator receives 415 J of beat energy from the reservoir at 270 K, how...
-
A refrigerator is removing heat from a cold medium at 3C at a rate of 7200 kJ/h and rejecting the waste heat to a medium at 30C. If the coefficient of performance of the refrigerator is 2, the power...
-
You are given 250 g of coffee (same specific heat as water) at 80.0C (too hot to drink). In order to cool this to 60.0C, how much ice (at 0.0C) must be added? Ignore heat content of the cup and heat...
-
How do you ensure data quality?
-
What are the guidelines on selecting and changing accounting policies?
-
Consider the situation in Merton's jump-diffusion model where the underlying asset is a non-dividend-paying stock. The average frequency of jumps is one per year. The average percentage jump size is...
-
Refer to the bond situation of Hewitt Corporation in Exercise 15-27. Hewitt issued the bonds at the advertised price. The company uses the straight-line amortization method and reports financial...
-
Find the frequency of small oscillations for a thin homogeneous plate if the motion takes place in the plane of the plate and if the plate has the shape of an equilateral triangle and is suspended...
-
As Michael O'Leary reflected on his two decades at the helm of Ryanair, he was pleased with the results of his strategy for the airline. When he assumed the position of CEO in 1994, Ryanair was a...
-
Senior Home Living (SHL) is a Canadian-based corporation located in British Columbia. SHL provides senior living residences across Canada. The company was incorporated in 1975, and has been...
-
In 2000, Middle, Inc. acquired 60 percent of Topper Company's common stock at a cost equal to Middle's share of Topper's nbv. For 2001, Middle and Topper reported the following: Separate Income (from...
-
Whitmer Corporation is working on its direct labor budget for the next two months. Each unit of output requires 0.07 direct labor-hours. The direct labor rate is $8.70 per direct labor-hour. The...
-
A mass of 3kg is free to move on a horizontal frictionless surface and attached to a spring of k=15 N/m. It is displaced from equilibrium by 0.1m to the right and released from rest. a. What is the...
-
A company had the following assets and liabilities at the beginning and end of this year. Beginning of the year End of the year Assets $ 58,000 103,000 Liabilities $ 24,865 41,715 a. Owner made no...
-
Mason Company provided the following data for this year: Sales Direct labor cost Raw material purchases Selling expenses Administrative expenses Manufacturing overhead applied to work in process...
-
A 0.29 kg mass is attached to the end of a spring that is attached horizontally to a wall. When the mass is displaced 10.2 cm and then released, it undergoes SHM. The force constant of the spring is...
-
The cost accountant for Sunset Fashions has compiled the following information for last quarter's operations: Administrative costs Merchandise inventory, April 1 Merchandise inventory, June 30...
-
1. What is the semi-annually compounded interest rate if $200 accumulates to $318.77 in eight years? Answer in percentage with two decimal places. 2. What is the quarterly compounded interest rate if...
-
Amino acids can act as ligands toward transition metal ions. The simplest amino acid is glycine (NH2CH2CO2H). Draw a structure of the glycinate anion (NH2CH2CO2-), acting as a bidentate ligand. Draw...
-
Describe how you could separate the ions in each of the following groups by selective precipitation. a. Ag+, Mg2+, Cu2+ b. Pb2+, Ca2+, Fe2+ c. Cl-, Br-, I- d. Pb2+, Bi3+
-
A sample of nitrogen gas was collected over water at 20. o C and a total pressure of 1.00 atm. A total volume of 2.50 10 2 mL was collected. What mass of nitrogen was collected? (At 20. o C the...
-
Show from combination of the equation for current and the flux expression that the conductivity of an electrolyte solution is given by \[\kappa=\frac{F^{2}}{R_{\mathrm{G}} T} \sum_{i}^{N} D_{i}...
-
Verify by inputting the various constants that \(R_{\mathrm{G}} T / F\) is equal to \(k_{\mathrm{B}} T / e\).
-
Consider an uncharged membrane of thickness \(100 \mu \mathrm{m}\) with concentrations of \(1 \mathrm{M} \mathrm{HCl}\) on one side and \(0.1 \mathrm{M} \mathrm{HCl}\) on the other side. Assume that...

Study smarter with the SolutionInn App


