Calculate the number of tetrahedral atoms per nm 3 for each zeolite framework type listed in the
Question:
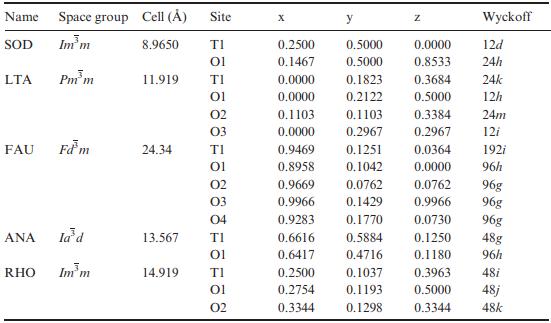
Calculate the number of tetrahedral atoms per nm3 for each zeolite framework type listed in the table below.
Transcribed Image Text:
Name Space group Cell () SOD Imm LTA FAU ANA RHO Pmm Fdm la'd Im m 8.9650 11.919 24.34 Site TI 01 14.919 TI 01 02 03 TI 01 02 03 04 13.567 TI 01 TI 01 02 X 0.2500 0.5000 0.1467 0.5000 0.0000 0.1823 0.0000 0.2122 0.1103 0.0000 0.9469 0.8958 0.9669 0.9966 0.9283 y 0.2500 0.2754 0.3344 0.1103 0.2967 0.1251 0.1042 0.0762 0.1429 0.1770 0.6616 0.5884 0.6417 0.4716 0.1037 0.1193 0.1298 Z 0.0000 0.8533 0.3684 0.5000 0.3384 0.2967 0.0364 0.0000 0.0762 0.9966 0.0730 0.1250 0.1180 0.3963 0.5000 Wyckoff 12d 24h 24k 12h 24m 12i 192i 96h 96g 96g 96g 48g 96h 48i 48j 0.3344 48k
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
See table Note cells are for the framework type such that T per nm ...View the full answer

Answered By

GERALD KAMAU
non-plagiarism work, timely work and A++ work
4.40+
6+ Reviews
11+ Question Solved
Related Book For 

Solid State Materials Chemistry
ISBN: 9780521873253
1st Edition
Authors: Patrick M. Woodward, Pavel Karen, John S. O. Evans, Thomas Vogt
Question Posted:
Students also viewed these Sciences questions
-
Zeolite A can be described using a 11.9 primitive cubic cell with the origin at a -cage centerin Figure 14.3. From the figure, make a visual estimate of the size of a sphere that would fit inside...
-
The -cristobalite polymorph of SiO 2 has a tetragonal cell with a = 4.971 , b = 6.922 , with Si on 2a and O on 4f Wyckoff sites. The SiO bond length is 1.60 and the average OO distance within...
-
Use the accompanying graph of y = f(x). Does exist? If it does, what is it? lim f(x)
-
Using the Hund rules, find the magnetic moment of the ground state of the atom whose open sub shell is half-filled with five electrons.
-
Find, in the form u + iv, sin 2i
-
When the Concepcions went to AT&T to get a phone, they were confused as to why they were paying money for the phone when it was advertised as being free. They had to pay sales tax on the full normal...
-
At the beginning of the current season on April 1, the ledger of Thousand Oaks Pro Shop showed Cash $2,500; Inventory $3,500; and Common Stock $6,000. The following transactions were completed during...
-
17. In many cases, a worker's hourly wage depends on the number of hours worked. If a certain number of hours is reached, the pay rate beyond that point increases. When working beyond a certain...
-
The table above contains ideal framework coordinates for different zeolites. Use a package such as vesta (http://jp-minerals.org/vesta/en/) to produce a 3D drawing of each structure. For each example...
-
Calculate the surface area of (a) A 1 g single crystal of cristobalite (density = 2.3 g/cm 3 , assume the crystal takes a spherical shape), (b) A uniform powder of spherical crystals each with a...
-
Wilhelms Cola Company plans to market a new lime-flavored cola this summer. The decision is whether to package the cola in returnable or in nonreturnable bottles. Currently, the state legislature is...
-
How do group cohesiveness and team composition affect the innovation process in cross-functional project teams? Explain
-
Units of output 1,200 units Price per unit $ 150 Variable cost per unit 90 Total fixed costs 48,000 Required: Determine the following: 1. Quarterly operating profit when 1,200 units are sold....
-
Innovation intermediate! You lack the impassioned stargazing of the innovation impresario. But you're not exactly the eyes-on-the-road, hands-on-the-wheel type either. It's crossed your mind, every...
-
Fifteen years ago a couple purchased a house for $190,000.00 by paying a 20% down payment and financing the remaining balance with a 30-year mortgage at 6.04% compounded monthly. (a) Find the monthly...
-
2. Titania Co. sold a 12% bond worth $600,000 on June 1, 2015. The bond pays interest on December 1 and June 1. The bond's maturity date is June 1, 2020. The bond yields 10%, sold for $638,780. On...
-
Hartford Mutual Insurance Co. issued a check for $60,150 payable to Andrew Michael Bogdan, Jr., Crystal Bogdan, Oceanmark Bank FSB, Goodman-Gable-Gould Company. The check was to pay a claim related...
-
on 8 For the following set of lengths 130, 170, 160, 160, 150, 190 Third quartile is: et red d out of Select one: O a. 160 a question O b. 145 O c. 175 O d. 180
-
Predict the final product(s) for each sequence of reactions: (a) (b) (c) (d) Br Br. 1) Excess NaNH, 2) EECI 3) H2, Lindlar's catalyst 1) NANH, 2) Mel 3) 9-BBN 4) H202, NaOH H-CEC-H
-
When (R)-4-bromohept-2-yne is treated with H 2 in the presence of Pt, the product is optically inactive. Yet, when (R)-4-bromohex-2-yne is treated with the same conditions, the product is optically...
-
If the reaction Fe 2 N(s) + 3/2H 2 (g) 2Fe(s) + NH 3 (g) comes to equilibrium at a total pressure of 1 bar, analysis of the gas shows that at 700. and 800. K, PNH 3 /PH 2 = 2.165 and 1.083,...
-
Meteor, Inc. recognized revenue of $ 6 0 0 for the current year, of which $ 1 2 0 was received in cash. The rest of the cash will be collected in the following year. The company incurred expenses of...
-
A customer in Halifax purchased a computer and paid $ 2 0 1 . 0 0 in HST ( 1 5 % ) . What was the total cost including HST of the computer purchase?
-
Recognizing that fixed costs are expected to increase, project a 2 0 % increase in fixed costs over last month, and determine a new break - even point in units for each of the three products. How...

Study smarter with the SolutionInn App


