The images below show a portion of a chain of the conducting polymer polypyrrole, before (top) and
Question:
The images below show a portion of a chain of the conducting polymer polypyrrole, before (top) and after (bottom) oxidative doping.
(a) Is the charge carrier formed in this process a polaron, bipolaron, or soliton?
(b) Is the conducing polymer that results doped n- or p-type?
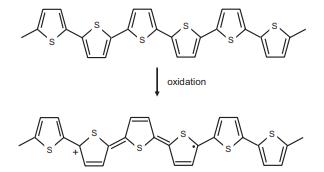
Transcribed Image Text:
oxidation S
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
a A single carbocationradical ...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Solid State Materials Chemistry
ISBN: 9780521873253
1st Edition
Authors: Patrick M. Woodward, Pavel Karen, John S. O. Evans, Thomas Vogt
Question Posted:
Students also viewed these Sciences questions
-
Managing Scope Changes Case Study Scope changes on a project can occur regardless of how well the project is planned or executed. Scope changes can be the result of something that was omitted during...
-
Planning is one of the most important management functions in any business. A front office managers first step in planning should involve determine the departments goals. Planning also includes...
-
Company History. Wawa Wild Wings, (WWW) was formed in 1980 by 3 residents of the town of Wawa Ontario, located about 230 kilometers north of Sault Ste. Marie Ontario. The 3 equal partners are Cosmo...
-
The September 30, 2018, adjusted trial balance of Buzzy?s, Inc., is shown next. Requirements 1. Prepare the September closing entries for Buzzy?s, Inc. 2. Calculate the ending balance in Retained...
-
The mass of 0 can be measured by observing the reaction + p 0 + n at very low incident kinetic energy (assume it is zero). The neutron is observed to be emitted with a kinetic energy of 0.60MeV....
-
Implement a method with signature concatenate(LinkedQueue Q2) for the LinkedQueue class that takes all elements of Q2 and appends them to the end of the original queue. The operation should run in...
-
Consider the following cash flow profile and assume MARR is 10 percent/year. a. What does Descartes' rule of signs tell us about the IRR(s) of this project? b. What does Norstrom's criterion tell us...
-
Disposal of assets. Answer the following questions. 1. A company has an inventory of 1,100 assorted parts for a line of missiles that has been discontinued. The inventory cost is $78,000. The parts...
-
Find the missing side lengths (in kilometers). (The sketches are not to scale.) 33 km 24 km 48 89 x 15 km 48% 89 22 km X = y = km E E km
-
Consider the electronic structure of graphene. (a) At how many points in the first Brillouin zone do the conduction and valence bands of graphene meet? (b) What is the name given to those points? (c)...
-
Each of the following compounds contains an octahedrally coordinated transition metal ion and behaves as an insulator/semiconductor. Identify each as either a band insulator or a MottHubbard...
-
The right of all partners to contract for a partnership is called a . Mutual agency. b . A partnership. c . A partners agreement. d . Voting rights. e . None of these answers
-
Tim Trepid is highly risk-averse while Mike Macho actually enjoys taking a risk. Investments Returns: Expected Value Standard Deviation Buy stocks $ Buy bonds Buy commodity futures Buy options 9,700...
-
1. Why do you think Everett set such an ambitious goal for transforming its fleet? Based on its achievements so far, do you think it is likely to meet that goal? Why or why not? 2. What additional...
-
identify three to five of the most important turning points in your career that have led to where you are now- serendipitous moments or opportunities that turned your career in another direction....
-
1. Write about a experiences working as a leader and/or with a leader: try to describe your experience according to trait, situational and functional perspectives. How might these different ways of...
-
Identify and give examples to illustrate the following aspects of customer relationship management, enterprise resource management, and supply chain management systems: a. Business processes...
-
Critics of the CAFA argue that the legislation deprive Americans of their day in court when they are wronged by powerful corporations. Under what circumstances could this criticism be justified?
-
Describe a group you belong or have belonged discuss the stages of group development and suggest how to improve the group effectiveness by using the group development model.
-
From the following data at 298.15 K as well as data in Table 4.1 (Appendix B, Data Tables), calculate the standard enthalpy of formation of H 2 S(g) and of FeS 2 (s): AR(kJ mol) Fe(s) + 2H2S(g) ...
-
Which of Ne or Ar has the larger van der Waals parameter b? Explain your reasoning.
-
You have calculated the pressure exerted by ethane using the ideal gas law and the RedlichKwong equations of state. How do you decide if the repulsive or attractive part of the molecular potential...
-
Select any accounts that would be included on the Income Statement of a company (scroll down to see all choices): Check All That Apply DividendsDividends Accounts PayableAccounts Payable Rent...
-
a. The best tax planning strategy for Sally is to make the contribution in Y2 b. If rates are increasing in the future, it is best to accelerate deductions c. The best tax planning strategy for Sally...
-
1. This is a treasure hunt on the tax forms. Find the form and the line of the form that each item for Rodger and Jessica Rabbit would appear on FORM LINE Jessica Rabbit's Income Rodger's income from...

Study smarter with the SolutionInn App


