1 H NMR spectra C through F (see below) correspond to four isomeric alcohols with the mole...
Question:
1H NMR spectra C through F (see below) correspond to four isomeric alcohols with the mole cular formula C5H12O. Try to assign their structures.
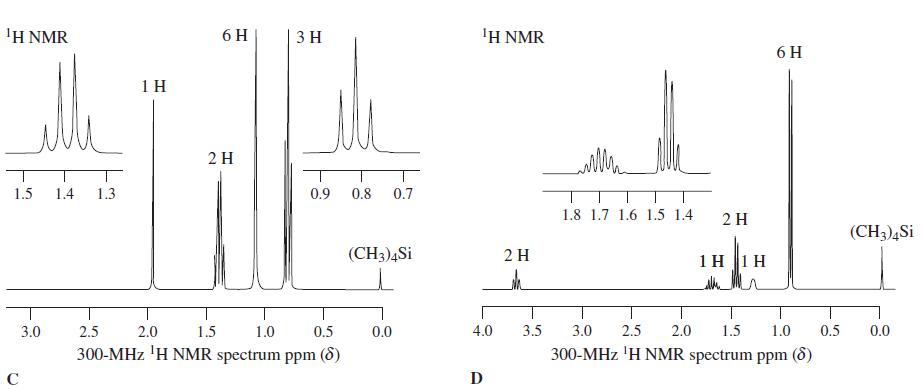
Transcribed Image Text:
1Η ΝMR 6 H 3 H 'H NMR 6 H 1 H 2 H 1.5 1.4 1.3 0.9 0.8 0.7 1.8 1.7 1.6 1.5 1.4 2 H (CH3)4Si (CH3)4Si 2H 1 H 1 H 3.0 2.5 2.0 1.5 1.0 0.5 0.0 4.0 3.5 3.0 2.5 2.0 1.5 1.0 0.5 0.0 300-MHz 'H NMR spectrum ppm (d) 300-MHz 'H NMR spectrum ppm (8) C D
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 63% (11 reviews)
C In the spectra there is a peak which integrates 6 hydrogen which is singlet This means there is tw...View the full answer

Related Book For 

Organic Chemistry structure and function
ISBN: 978-1429204941
6th edition
Authors: K. Peter C. Vollhardt, Neil E. Schore
Question Posted:
Students also viewed these Sciences questions
-
Draw the structures of the four isomeric alcohols with molecular formula C4H9OH. (a) Which isomer has the 13C NMR spectrum shown in Figure 12.14? Explain why. (b) Two of the four isomers will have...
-
Propose structures for alcohols that have the following 1H NMR spectra: (a) C5H12O (b) C8H10O Part (a) TMS O ppm 10 3 2 Chemical shift (8) Part (b) TMS O ppm 10 8. 6. 4 3 2 Chemical shift (8) Inten
-
Propose structures for alcohols that have the following 1H NMR spectra: (a) C9H12O (b)C8H10O2 Part (a) TMS 10 O ppm Chemical shift (8) Part (b) TMS O ppm 10 8. Chemical shift (8) Intensity Intensity...
-
Consider the following data set that contains information about a sample of ten vehicles that recorded their gross weight and their fuel efficiency as stated by the manufacturer. Vehicle Vehicle...
-
What are the PMBOK defined processes for the procurement management knowledge area?
-
One acre of land is equivalent to 43,560 square feet. Write a program that calculates the number of acres in a tract of land with 389,767 square feet. Hint: Divide the size of the tract of land by...
-
There is increasing legislation encouraging employers to support work-life balance, but the emphasis is on family-friendly measures, and not on work-life balance for all. LO1
-
Let the expected pound return on a U.K. equity be 15%, and let its volatility be 20%. The volatility of the dollar/pound exchange rate is 10%. a. Graph the (approximate) volatility of the dollar...
-
Remo Company and Angelo Incorporated are separate companies that operate in the same industry. Following are variable costing income statements for the two companies showing their different cost...
-
Phillip and Case are in the process of forming a partnership to import Belgian chocolates, to which Phillip will contribute one-third time and Case full time. They have discussed the following...
-
Predict the spin spin splitting that you would expect to observe in the NMR spectra of each compound in Problem 37. CH3 CH3 CH3 (a) CH;CCH,CH3, BICH,CHCH,CH3, CH;CHCH,CH;Br Br CH,CI CH; (b)...
-
Sketch 1H NMR spectra for the following compounds. Estimate chemical shifts (see Section 10-4) and show the proper multiplets for peaks that exhibit spin spin coupling. (a) CH 3 CH 2 OCH 2 Br; (b)...
-
Once the pictures are stored on the Ohio companys email server, who owns the pictures? Who controls those pictures? Does the company have the right to inspect the contents of its employees mailboxes?...
-
If WHO, the World Health Organization,defines health as a state of completephysical, mental and social well-being and not merely the absenceof disease and infirmity (WHO, 2011)and wellness is...
-
As a manager, you want to find a way to motivate Nate and increase his engagement and job satisfaction in the workplace. Drawing upon a behavioral theory of motivation, discuss how you, as a manager,...
-
Why the sudden increase in income before taxes in 2021? 8. Why were the operating assets the highest in 2019? 9. Why are the short-term loans the highest in 2020? 10. Why are the other long-term...
-
Mercy wants to make sure that she will be able to provide for her daughter's college and plans to open a savings account with a bank that is ready to pay interest as shown below per year compounded...
-
Question 1. For a firm that uses portfolio management, please give a real or hypothetical example of how the CEO's personal bases for power help organizational performance. Question 2. Give a real...
-
Write a method called daysInMonth that takes a month (an integer between 1 and 12) as a parameter and returns the number of days in that month in this year. For example, the call daysInMonth(9) would...
-
Define relevant costs and discuss: (1) whether all future costs are relevant for decision making and (2) whether variable costs are always relevant and fixed costs are always irrelevant
-
Cylinders of compressed gas are typically filled to a pressure of 200 bar. For oxygen, what would be the molar volume at this pressure and 25C based on (a) The perfect gas equation, (b) The van der...
-
At 300 K and 20 atm, the compression factor of a gas is 0.86. Calculate (a) The volume occupied by 8.2 mmol of the gas under these conditions and (b) An approximate value of the second virial...
-
A vessel of volume 22.4 dm ' contains 1.5 mol H2 and 2.5 mol N2 at 273.15 K. Calculate (a) The mole fractions of each component, (b) Their partial pressures, and (c) Their total pressure.
-
Describe international business, including its various forms, and globalization. Explain the globalization debate and its effect on international business.
-
Please calculate Ke Cost of equity through CAPM Equity Beta 0.86 30-year Treasury bond yield, current 3.00% Treasury bill (3-month) yield, current 0.90% 30-y. Treasury bond yield, historical average...
-
Suppose the real interest rate is 1% and annual inflation is 2%. Calculate the annual nominal interest rate using the Fisher equation.

Study smarter with the SolutionInn App


