Question: How would each reaction in Problem 25 be affected by the addition of each of the following substances to the solvolysis mixture? (a) H 2
How would each reaction in Problem 25 be affected by the addition of each of the following substances to the solvolysis mixture?
(a) H2O
(b) KI
(c) NaN3
(d) CH3CH2OCH2CH3
In Problem 25
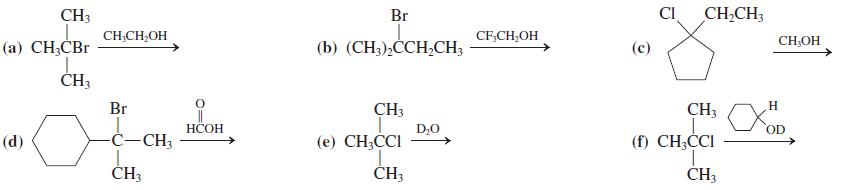
CH3 Br CI CH,CH; CH;CH,OH CF,CH;OH CH;OH () C,r (b) (CH).H-CH; () H3 Br CH3 CH3 H D;0 OD (d) --CH3 (e) CH;CCI (f) CH;CI CH3 H3 CH3
Step by Step Solution
★★★★★
3.45 Rating (158 Votes )
There are 3 Steps involved in it
1 Expert Approved Answer
Step: 1 Unlock

a This would result in a mixture of ether ... View full answer

Question Has Been Solved by an Expert!
Get step-by-step solutions from verified subject matter experts
Step: 2 Unlock
Step: 3 Unlock
Document Format (2 attachments)
970_61d57f260b869_827288.pdf
180 KBs PDF File
970_61d57f260b869_827288.docx
120 KBs Word File


