Give the major product(s) of the following reactions. Indicate which of the following mechanism(s) is in operation:
Question:
Give the major product(s) of the following reactions. Indicate which of the following mechanism(s) is in operation: SN1, SN2, E1, or E2. If no reaction takes place, write “no reaction.”
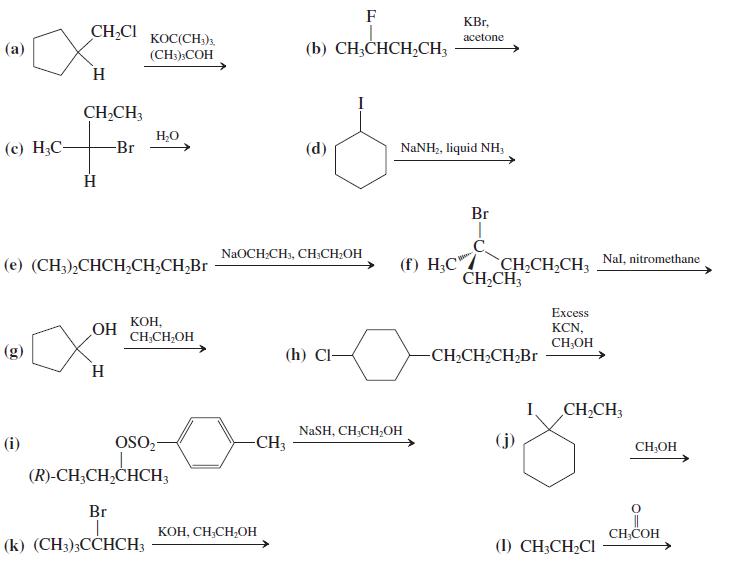
Transcribed Image Text:
F KBr, CH,CI KOC(CH;)3. аcetone (а) (b) CH;CHCH,CH3 (CH)»COH H. CH,CH3 H,0 (с) Н.С- -Br (d) NaNH2, liquid NH, Br C. CH CH,CH, Nal, nitromethane CH,CH3 NaOCH.CH, CH.CH.ОН (е) (CH3)-СНСH,CH,CH,Br (f) H;C" Excess OH KOH, CH,CH,OH KCN, CH;OH (h) Cl- -CH2CH2CH,Br H I. CH,CH3 NaSH, CH;CH;OH (i) OSO- -CH3 CH,OH (R)-CH;CH,CHCH3 Br KOH, CH;CH,OH CH;COH (k) (CH3)3CCHCH3 (1) CH3CH2CI
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (9 reviews)
a b C 1 CHC ...View the full answer

Answered By

Yadram Dhanka
I was engaged in conducting private tuitions for students of class 11th and 12th. I would like to work with a leading educational organization and to use my in-depth subject knowledge and passion towards teaching to the best of my ability, so as to enrich the student’s ability to learn, as well as to advance my career in the education sector.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry structure and function
ISBN: 978-1429204941
6th edition
Authors: K. Peter C. Vollhardt, Neil E. Schore
Question Posted:
Students also viewed these Sciences questions
-
Give the major product of each of the following reactions: a. b. c. d. e. f. HBr HC CCH3 peroxide excess Br 3 CH2Cl2 excesS CHC CCH HBr excess
-
Give the major product of each of the following reactions: a. b. c. d. e. f. g. h. HCI CH3OH H20 , H+ H20 HBr peroxide HBr Cl2 H2O Cl2 CH2CI
-
Give the major product of each of the following reactions a. b. c. d. e. f. g. h. i. CCH3 + HNO3 S NO2 CHs CH CHCH NCH CH 3 CH3 HO CH3NCH3 + PC15- NC 1. H202 2. CH3 1. HO 3. Ht CH3 + CH:CH2MgBr 2.4...
-
The Bay City Parks and Recreation Department is considering building several new facilities, including a gym, an athletic field, a tennis pavilion, and a pool. It will base its decision on which...
-
The main goal of the quality assurance process is to do what?
-
Assume that in October you bought a $450 nonrefundable airline ticket to Telluride, Colorado, for a 5-day/4-night winter ski vacation. You now have an opportunity to buy an airline ticket for a...
-
Nereus Montemayor was an employee of VZ Hogs, a company that raises hogs and produces hog feed. VZ Hogs used an extruder manufactured by Sebright Products, Inc. to create hog feed out of discarded...
-
Jasmine Park encountered her boss, Rick Gompers, at the pop machine in the lobby. Rick is the vice president of marketing at Down South Lures Corporation. Jasmine was puzzled by some calculations she...
-
KORBIN COMPANY Comparative Balance Sheets December 31 Assets Current assets Long-term investments Plant assets, net Total assets Liabilities and Equity Current liabilities Common stock Other paid-in...
-
Let x be a random variable that represents the percentage of successful free throws a professional basketball player makes in a season. Let y be a random variable that represents the percentage of...
-
How would each reaction in Problem 25 be affected by the addition of each of the following substances to the solvolysis mixture? (a) H 2 O (b) KI (c) NaN 3 (d) CH 3 CH 2 OCH 2 CH 3 In Problem 25 CH3...
-
Consider the general substitution-elimination reactions of the bromoalkanes. How do the reaction mechanisms and product formation differ when the structure of the substrate and reaction conditions...
-
Wal-Mart Stores, Inc., is the world's largest retailer. A large portion of the premises that the company occupies are leased. Its financial statements and disclosure notes revealed the following...
-
Can the concept of moral panics, as theorized by Cohen and others, help explain the process through which media, politicians, and moral entrepreneurs construct and amplify public anxieties about...
-
Margot bought a small residential rental building 15 years ago for $150,000. In the current year, she sold it for $400,000. She had taken $55,000 of straight-line depreciation. Margot is in the 35...
-
How do evolutionary psychology perspectives contribute to our understanding of the adaptive functions and evolutionary origins of gendered behaviors, roles, and preferences, and what are the...
-
How the contract and record keeping should be done by supervisor while assisting supervisor? Explain briefly
-
In reviewing an income statement, you notice an operating income of $52,500 and an income tax rate of 25% of income. If you are looking for the amount to report for income for financial accounting...
-
How should a company handle LIFO liquidation in an interim period when the liquidated inventory is expected to be replaced by year-end?
-
How has the globalization of firms affected the diversity of their employees? Why has increased diversity put an additional burden on accounting systems?
-
Suppose that 2.5 mmol Ar (g) occupies 72 dm3 at 298 K and expands to 100 dm3, Calculate G for the process.
-
Diamond, an allotrope of carbon, is the hardest substance and the best conductor of heat yet characterized. For these reasons, diamond is used widely in industrial applications that require a strong...
-
The change in the Gibbs energy of a certain constant-pressure process was found to fit the expression G/T =-73.1 + 42.8(T/K). Calculate the value of S for the process.
-
In 2023, Mary Jane and her husband Peter, both architects, come to your accounting firm to seek help. They tell you, "We are concerned we're paying more tax than we owe. Our last accountant said we...
-
look up the cost of copper and the cost of zinc. How much is one penny worth in raw material?
-
What is tax planning? define its impacts and how it was implimented?

Study smarter with the SolutionInn App


