Name the following alcohols according to the IUPAC nomenclature system. Indicate stereochemistry (if any) and label the
Question:
Name the following alcohols according to the IUPAC nomenclature system. Indicate stereochemistry (if any) and label the hydroxy groups as primary, secondary, or tertiary.
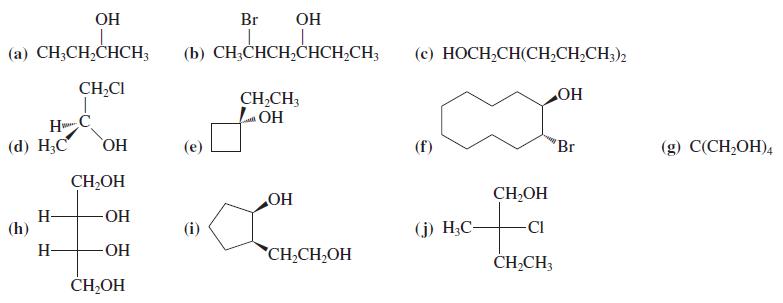
Transcribed Image Text:
OH Br OH (a) CH;CH,CHCH; (b) CH;CHCH,CHCH,CH; (c) HOCH,CH(CH,CH,CH;)2 CH,CI CH,CH, LOH HO H (d) Н,С HO CH,OH (f) 'Br (g) С(CH,ОH), CH,OH H- (h) HO OH (i) (j) H;C- CI H- -ОН CH,CH,OH ČH,CH; CH-OH
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (10 reviews)
SNo IUPAC Name Type Of Alcohol Stereochemistry a Butan2ol secondary NA b 2bromohexan4ol s...View the full answer

Answered By

Bharat Singh Patel
I like teaching since my matriculation. That time i was teaching 6-8 class students with all subjects. I was very interested in teaching. Then after i have done my 10+2. I joined a coaching class. Where i was teaching mathematics and chemistry of 10th class student. After that i moved to Ewing Christian College, Allahabad for Graduation. then i taught chemistry only. then I clear IIT JAM to got admission in IIT Jodhpur for M.Sc. During M.Sc I have cleared CSIR-JRF With AIR 54 and GATE with AIR 114. In college time I always cleared doubts of classmates and juniors.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry structure and function
ISBN: 978-1429204941
6th edition
Authors: K. Peter C. Vollhardt, Neil E. Schore
Question Posted:
Students also viewed these Sciences questions
-
Classify the following alcohols as primary, secondary, or tertiary: (a) (b) (c) (d) OH
-
State whether each of the following alcohols is primary, secondary, or tertiary. a. b. c. d. -, ,
-
Classify the following amines as primary, secondary, or tertiary: (a) (b) (c) (d) (e) (f) H IN NH2 HN
-
Suppose that General Motors Acceptance Corporation issued a bond with 10 years until maturity, a face value of $1000, and a coupon rate of 7% (annual payments). The yield to maturity on this bond...
-
Describe the importance of managing stakeholder expectations.
-
A five-star hotel buys bouquets of flowers to decorate its common areas and guest rooms. Its flexible budget for flowers is $325 per day of operations plus $7.20 per room-day. (A room-day is a room...
-
Amanda Forbes was hired as a nail technician by Showmann, Inc., in 2011. In 2017, Forbes attended a work-related holiday party where Showmann distributed raffle tickets to employees. One of the...
-
Braun Company produces its product through a single processing department. Direct materials are added at the beginning of the process. Direct labor and overhead are added to the product evenly...
-
What is a sampling distribution? Explain
-
Determine the force P that must be applied perpendicular to the handle in order to hold the mechanism in equilibrium for any angle of rod CD. There is a couple moment M applied to the link BA. A...
-
Which of the following isomeric carbocations is the most stable? CH,+ CH3 () (b) -CH3 CH3 () (d)
-
Write all products of the reaction of each of the alcohols in Problem 31 with concentrated H 2 SO 4 under elimination conditions. In Problem 31 OH () CH-CH-CH,O (b) CH;CHCH3 (c) CH;CH,CH,CH,OH OH (d)...
-
Which of the following subsets are independent? Support your answer. (a) {[1 -1 O]T, [3 2 -1]T, [3 5 -2]T) in R3. (b) [(1 1 1]T, [1 -1 l]T, [0 0 l]T} in R3. (c) {[1 -1 1 -1]T, [2 0 1 0]T, [0 -2 1...
-
What type of cost accounting system would be best suited for a company like Apple, INC.
-
Steph made a purchase on her credit card for $945. She had no previous balance, or amount unpaid from the month before. Her new balance, or amount now owed, is $945. She is only able to pay the...
-
In civil engineering, when vertical piles are subjected to lateral loads, what is the primary consideration? A ) Reduction of soil erosion B ) Prevention of water contamination C ) Stability and...
-
A borrower's monthly mortgage payment is $1,256.28. His annual property taxes are $809, and his annual insurance premium is $712. How much of his payment goes toward the principal and interest?
-
How do researchers effectively navigate the complexities of participant observation within the constructivist paradigm?
-
Mighty Company purchased a 60 percent interest in Lowly Company on January 1, 2012, for $420,000 in cash. Lowly's book value at that date was reported as $600,000 and the fair value of the...
-
What types of inventory issues Starbucks might reflect upon at the end of each year? The mission of Starbucks is to inspire and nurture the human spiritone person, one cup, and one neighborhood at a...
-
Calculate the difference in molar entropy (a) between liquid water and ice at -5C, (b) between liquid water and its vapour at 95C and 1.00 atm. The differences in heat capacities on melting and on...
-
A block of copper of mass 2.00 kg (Cp, m = 24.44 T K-I mol-1) and temperature OC is introduced into an insulated container in which there is 1.00 mol H20 (g) at 100C and 1.00 atm. (a) Assuming all...
-
At 20C, the density of a 20 per cent by mass ethanol-water solution is 968.7 kg m-3. Given that the partial molar volume of ethanol in the solution is 52.2 cm3 mol-1, calculate the partial molar...
-
T is a C corporation and a calendar year taxpayer. T has gross receipts of $ 2 7 million in Year 2 0 2 1 , $ 2 8 million in Year 2 0 2 2 , and $ 3 8 million in Year 2 0 2 3 . T is not required to add...
-
Crane Co. has annual obligations of $30 toward interest and principal and is forecasting a cash flow of either $25 (Scenario I) or $50 (Scenario 2) in the coming year. What will the payment to...
-
Write a Python script to simulate Monte Carlo method using numpy library.

Study smarter with the SolutionInn App


