What are the products (and their ratios) of periodic acid cleavage of each of the following substances:
Question:
What are the products (and their ratios) of periodic acid cleavage of each of the following substances:
(a) 1,3-dihydroxyacetone;
(b) rhamnose (Problem 34);
(c) glucitol.
Problem 34
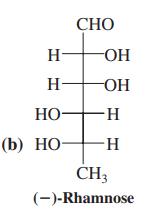
Transcribed Image Text:
СНО H- OH H OH НО H- (b) НО- -H- CH3 (-)-Rhamnosе
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 44% (9 reviews)
a b c HO 13 ...View the full answer

Answered By

Anurag Verma
I am a master in MSc chemistry with specialization in organic chemistry. I qualify national labvel exam of India like CSIR-JRF and GATE with good rank. I also passed various IIT interview of India for the admission in PhD. I also part time teacher and teach online and offline to a student of chemistry. I can solve any problem of organic chemistry also solve paper and teach spectroscopy, organomatellic chemistry, Inorganic chemistry also.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry structure and function
ISBN: 978-1429204941
6th edition
Authors: K. Peter C. Vollhardt, Neil E. Schore
Question Posted:
Students also viewed these Sciences questions
-
Predict the products formed by periodic acid cleavage of the following diols. (a) CH3CH(OH)CH(OH)CH3 (b) (c) (d) CHAOH OH OH Ph-CCH OH)CH,CH CH HO HO
-
What are the products of the following reaction? (a) C 6 H 5 I + CH 3 OH (b) C 6 H 5 OH + CH 3 I (c) C 6 H 5 I + CH 3 I (d) CH;OCH, HL A, HI. A, ?
-
What are the products of reaction of (E)-3-methyl-3-hexene with each of the reagents in Problem 46? (a) H 2 , PtO 2 , CH 3 CH 2 OH (b) D 2 , Pd C, CH 3 CH 2 OH (c) BH 3 , THF then NaOH + H 2 O 2 (d)...
-
ABC Company produces and sells I product. Once the products are produced, they are sold, and there is no work-in- process, no any inventory in stock. Company uses standard costing method in its...
-
Use the data in WAGEPRC.RAW for this exercise. Problem 11.5 gave estimates of a finite distributed lag model of gprice on gwage, where 12 lags of gwage are used. (i) Estimate a simple geometric DL...
-
When a parent-subsidiary relationship exists, consolidated financial statements are prepared in recognition of the accounting concept of a. Reliability. b. Materiality. c. Legal entity. d. Economic...
-
Renkas Heaters selected data for October 2017 are presented here (in millions): Calculate the following costs: 1. Direct materials inventory 10/31/2017 2. Fixed manufacturing overhead costs for...
-
On January 1, 2012, Tiggy Company purchased the following two machines for use in its production process. Machine A: The cash price of this machine was $38,000. Related expenditures included: sales...
-
Using Doug Melville's checklist from his TEDx Talk evaluate which of his recommendations for improving diversity awareness are most useful to business leaders, and why.
-
John and Sandy Ferguson got married eight years ago and have a seven-year old daughter Samantha. In 2014, John worked as a computer technician at a local university earning a salary of $52,000, and...
-
Ketoses show positive Fehlings and Tollenss tests not only by oxidation to -dicarbonyl compounds, but through a second process: Ketoses isomerize to aldoses in the presence of base. The aldose then...
-
Write the expected products of the reaction of each of the following sugars with (i) Br 2 , H 2 O; (ii) HNO 3 , H 2 O, 60C; (iii) NaBH 4 , CH 3 OH; and (iv) excess C 6 H 5 NHNH 2 , CH 3 CH 2 OH, ....
-
If a = 4 m, determine the magnitudes of P and Q required to maintain the cable in the shape shown. DI 120 kN
-
A series, or stream, of cash flows may not always necessarily be an annuity. Cash flows can also be uneven and nonconstant, but the concept of the time value of money applies to uneven cash flows as...
-
Who was Marie Tagliofil and what did she contribute to the To" century Romantic era Of ballet? (Video. Romantic Era- Marie Taglioni 1804-1884) What was ballet dancer Anna Pavlova known for? How has...
-
Arrow wants to buy a new item of equipment which will be used to provide a service to customers of the company. Two models of equipment are available, one with a slightly higher capacity and greater...
-
What types of messages would you personally need extra elaboration on to sway you more one way or another? Out of the 5 influences on motivation, which one would be more effective when trying to...
-
Which kind of ad would you have been most likely to click? Was this because the ad was superior or simply because it matched your idiosyncratic preferences better? (If there is only one ad, analyze...
-
Jackson uses his automobile 90% for business and during 2014 drove a total of 14,000 miles. Information regarding his car expenses is listed below. Business parking...
-
Privitera and Freeman (2012) constructed a scale to measure or estimate the daily fat intake of participants; the scale was called the estimated daily intake scale for fat (EDIS-F). To validate the...
-
What aromatic products would you obtain from the KMnO4 oxidation of the followingsubstances? (a) O2N. (b) C(CH3)3 CH(CH3)2 "
-
Refer to Table 5.3 for quantitative idea of the stability of a benzyl radical. How much more stable (in kJ/mol) is the benzyl radical than a primary alkyl radical? How does a benzyl radical compare...
-
Styrene, the simplest alkenylbenzene, is prepared commercially for use in plastics manufacture by catalytic dehydrogenation of ethylbenzene. How might you prepare styrene from benzene using reactions...
-
The purpose of this project is to demonstrate your grasp of the concepts you have learned throughout the course and your ability to analyze the financial standings and history of a company. You...
-
Why do department managers track direct costs within their departments?
-
Based on the bank's DCR (and assuming operating expenses are $450,000), what is the maximum allowable annual debt service (rounded to the nearest hundred)?

Study smarter with the SolutionInn App


