Write all sensible products of the reaction of each of the alcohols in Problem 31 with concentrated
Question:
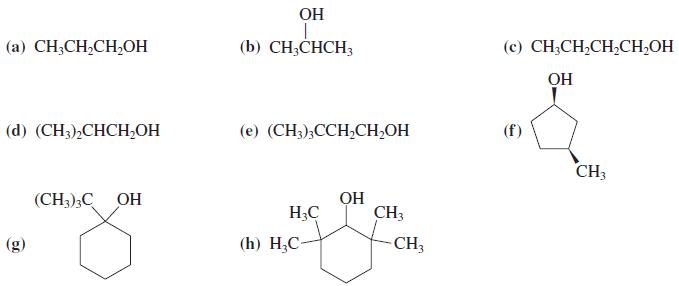
Write all sensible products of the reaction of each of the alcohols in Problem 31 with concentrated aqueous HBr.
In Problem 31
Transcribed Image Text:
OH (а) CH-CH-CH,Oн (b) CH;CHCH3 (c) CH;CH,CH,CH,OH OH (d) (CH),СНCН,ОН (е) (CH),ССH-CH-OH CH3 (CH3);C OH OH H3C CH3 (g) (h) H,С- CH3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 71% (7 reviews)

Answered By

Astha Sharma
I have cleared many exams like GATE, CSIR-NET. Currently i am a lecturer of chemistry. I have an experience of 4 years in teaching.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Organic Chemistry structure and function
ISBN: 978-1429204941
6th edition
Authors: K. Peter C. Vollhardt, Neil E. Schore
Question Posted:
Students also viewed these Sciences questions
-
Write all products of the reaction of each of the alcohols in Problem 31 with concentrated H 2 SO 4 under elimination conditions. In Problem 31 OH () CH-CH-CH,O (b) CH;CHCH3 (c) CH;CH,CH,CH,OH OH (d)...
-
Give the expected main product of the reaction of each of the alcohols in Problem 31 with PBr 3 . Compare the results with those of Problem 33.
-
Write the expected products of the reaction of each of the following sugars with (i) Br 2 , H 2 O; (ii) HNO 3 , H 2 O, 60C; (iii) NaBH 4 , CH 3 OH; and (iv) excess C 6 H 5 NHNH 2 , CH 3 CH 2 OH, ....
-
During one week, four production employees of Martinez Manufacturing Company worked the hours shown below. All these employees receive overtime pay at one and one-half times their regular hourly rate...
-
List the principle goals of team-building activities.
-
Budgeted unit sales for March, April, and May are 75,000, 80,000, and 90,000 units. Management desires to maintain an ending inventory equal to 30% of the next months unit sales. How many units...
-
SWIGART v. BRUNO CALIFORNIA COURT OF APPEALS 13 CAL. APP. 5TH 529 2017 According to the American Endurance Ride Conference, endurance riding is a highly competitive and demanding sport. It is...
-
Some special handling devices can be obtained for $12,000. At the end of 4 years, they can be sold for $600. Compute the depreciation schedule for the devices using the following methods: (a)...
-
Define the general - purpose financial statements and points out the limitation of such statements.
-
SPG is a US MNC that has operations in Europe, the Middle East, and Africa. It has identified needs of leverage of USD24 million in 7 months for a six-month period. To lock in the rate on this loan,...
-
Name the following alcohols according to the IUPAC nomenclature system. Indicate stereochemistry (if any) and label the hydroxy groups as primary, secondary, or tertiary. OH Br OH (a) CH;CH,CHCH; (b)...
-
Give detailed mechanisms and final products for the reaction of 3-methyl-2-pentanol with each of the reagents that follow. (a) NaH (b) Concentrated HBr (c) PBr 3 (d) SOCl 2 (e) Concentrated H 2 SO 4...
-
Tell how many units and in what directions the graphs of the given equations are to be shifted. Give an equation for the shifted graph. Then sketch the original and shifted graphs together, labeling...
-
How does the phenomenological approach in qualitative research facilitate a deeper understanding of lived experiences?
-
Your grandpa met a stock broker at the supermarket who convinced him to spend $10K on Tesla stocks. This stock broker does have a valid license from the government. Still, what are the things that...
-
Check Each Of Following , - is a decreasing sequence; that is, show that 1. Show that the sequence 2 3 4 5 6 7 - - - 1'2'3'4'5'6' (n + 1) term in the sequence is less than the nth term.
-
1) h C b 2) S S b = 9.17 mm a = 4.4 mm c = 9.8 mm h = 4.1 mm Area: Perimeter: Type: s = 5.5 yds h = 4.8 yds Area: Perimeter: Type: 5) 4) b a h C S S S h S a = 5.81 cm b = 8.48 cm c = 9.3 cm h = 5.2...
-
Sam Inc. is a 90% owned subsidiary of Paul Corp. Paul sold land to Sam for $100,000 that originally cost Paul $50,000. Paul uses the fully adjusted equity method. What adjustment is needed on Paul's...
-
Up and its 80 percent owned subsidiary (Down) reported the following figures for the year ending December 31, 2013. Down paid dividends of $30,000 during this period. In 2012, unrealized gross...
-
Juarez worked for Westarz Homes at construction sites for five years. Bever was a superintendent at construction sites, supervising subcontractors and moving trash from sites to landfills. He...
-
Predict the partial vapour pressure of the component B above its solution in A in Exercise 5.3b when the molalityofB is 0.25 mol kg-I. The molar mass of A is 74.1 g mol-1.
-
Discuss what would be observed as a sample of water is taken along a path that encircles and is close to its critical point.
-
The vapour pressure of2-propanol is 50.00 kPa at 338.8C, but it fell to 49.62 kPa when 8.69 g of an in volatile organic compound was dissolved in 250 g of 2-propanol. Calculate the molar mass of the...
-
3. If tolerable misstatement as percentage of 630,000 population value is 5% and and Confidence factor is 3.68 Calculate sampling interval (1 Point) 8560 31500 63.7 73.6 4. Increasing ARIA leading to...
-
List 3 factors that officers rely on to evaluate H&C applications.?
-
On its income statement for the year, Crescent Ltd. reported $100,000 of selling and administrative expenses, which included $20,000 in depreciation expense. During the year prepaid expenses...

Study smarter with the SolutionInn App


