a. Name each of the following alcohols. b. Name each of the following alcohols, including the stereochemistry
Question:
a. Name each of the following alcohols.
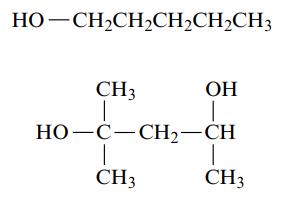
b. Name each of the following alcohols, including the stereochemistry if cis–trans isomers are possible.
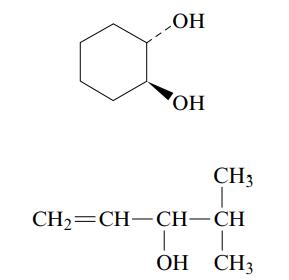
Transcribed Image Text:
HỌ—CH,CH,CH,CH,CH3 ОН I CH3 T HỌ–C–CH2–CH 1 CH3 CH3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 57% (7 reviews)
ANSWER Alcohols are substances in which any carbon atom in the main carbon chain has a hydroxyl grou...View the full answer

Answered By

Aketch Cindy Sunday
I am a certified tutor with over two years of experience tutoring . I have a passion for helping students learn and grow, and I firmly believe that every student has the potential to be successful. I have a wide range of experience working with students of all ages and abilities, and I am confident that I can help students succeed in school.
I have experience working with students who have a wide range of abilities. I have also worked with gifted and talented students, and I am familiar with a variety of enrichment and acceleration strategies.
I am a patient and supportive tutor who is dedicated to helping my students reach their full potential. Thank you for your time and consideration.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Chemistry
ISBN: 9781305957404
10th Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Question Posted:
Students also viewed these Engineering questions
-
Each of the following alcohols has been subjected to acidcatalyzed dehydration and yields a mixture of two isomeric alkenes. Identify the two alkenes in each case, and predict which one is the major...
-
Each of the following alcohols has been prepared by reaction of a Grignard reagent with ethylene oxide. Select the appropriate Grignard reagent in each case. (a) CH3 CH2CH2OH CH2CH20
-
Each of the following alcohols has been subjected to acidcatalyzed dehydration and yields a mixture of two isomeric alkenes. Identify the two alkenes in each case, and predict which one is the major...
-
Bruno Corp. has decided to expand its operations. The bookkeeper recently completed the following statement of financial position in order to obtain additional funds for expansion: Instructions (a)...
-
The Hughes Supply Company uses an inventory management method to determine the monthly demands for various products. The demand values for the last 12 months of each product have been recorded and...
-
Metro builds and operates traditional shopping malls. It holds a $25 million deferred tax asset relating to credit carryforwards at the state, local, and Federal levels. No valuation allowances...
-
What supplemental disclosures in notes to financial statements are required for investments in securities?
-
The following selected accounts and their current balances appear in the ledger of Case-It Co. for the fiscal year ended November 30, 2010: Instructions1. Prepare a multiple-step income statement.2....
-
Indicators are used in autoclaving to ensure that sterilization is complete. One type of chemical indicator turns color when it has reached a specific temperature; the other type turns color when it...
-
Balance a ration for a milking dairy cow to contain: CP = 16 % Ca = .66 % P= .41 % 27 % Corn Silage Feed Feed Feed 23 % Alfalfa 5 % Brewers Grains Feed 3 % Whole Cotton Seed Leave 2% slack space for...
-
Estradiol is a female hormone with the following structure: How many chiral carbon atoms are in estradiol? HO CH3 OH
-
The two isomers having the formula C 2 H 6 O boil at -23 C and 78.5 C. Draw the structure of the isomer that boils at -23 C and of the isomer that boils at 78.5 C.
-
Discuss the importance of a survey in the settlement procedure?
-
What is one way mass communication differs from other forms of communication?
-
Imagine that you recently became the president of your local Community Improvement Association, and you need to prepare a speech about the pros and cons of building a community swimming pool. Before...
-
Write a reflective paper on a significant interpersonal relationship in your life. it should discuss; Communication dynamics, challenges, and strategies for improving communication within that...
-
Choose two theories of economic development. illustrate the main concepts of each theory. Focusing on one of the theories, discuss how it helps us to understand the economic growth and development in...
-
How does the straight-line method calculate depreciation?
-
An object of mass 3.0 kg is projected into the air at a 55 angle. It hits the ground 3.4 s later. What is its change in momentum while it is in the air? Ignore air resistance.
-
The following cost information was provided to you for analysis: September 12,000 Units Produced Costs: TIC TAC TOE TING August 10,000 P80,000 70.000 60.000 50,000 How much is the fixed cost per...
-
Does the reduction of acetoacetyl ACP in step 6 occur on the Re face or the Si face of themolecule? NADPH NADP+ SACP SACP H3C Acetoacetyl ACP B-Hydroxybutyryl ACP
-
Identify the two steps in glycolysis in which ATP is produced.
-
Look at the entire glycolysis pathway and make a list of the kinds of organic reactions that take place-nucleophilic acyl substitutions, aldol reactions, E1cB reactions, and so forth.
-
Students must useinformation literacy in their writing. Theprojectshould be at least #2000 words of content. Word document only! The students will be required to meet with a top level manager,...
-
Your agency has been directed by community leaders to develop and implement a community paramedic program targeting injury prevention among the elderly. You are now tasked with providing a briefing...
-
Write up an assessment record for one of the children/young people in a learning environment. The record must be set out clearly showing: - a) Your assessment of the learner's development b) How the...

Study smarter with the SolutionInn App


