A Nb-W alloy held at 2800C is partly liquid and partly solid. (a) If possible, determine the
Question:
A Nb-W alloy held at 2800°C is partly liquid and partly solid.
(a) If possible, determine the composition of each phase in the alloy, and
(b) If possible, determine the amount of each phase in the alloy.
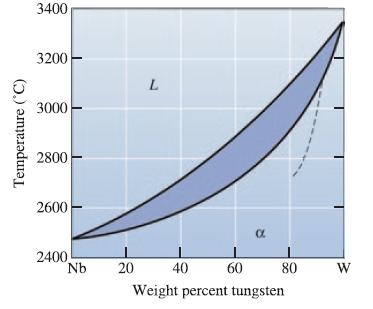
Transcribed Image Text:
Temperature (°C) 3400 3200 3000 2800 2600 2400 Nb L 20 α 40 60 Weight percent tungsten 80 W
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 28% (7 reviews)
a The composition of each phase in the al...View the full answer

Answered By

Brian Otieno
I'm Brian , an experienced professional freelancer with countless hours of success in freelancing many subjects in different disciplines. Specifically, I have handled many subjects and excelled in many disciplines. I have worked on many Computer Science projects and have been able to achieve a lot in that field. Additionally, I have handled other disciplines like History, Humanities, Social Sciences, Political science, Health care and life science, and Religion / Theology. My experience generally in these subjects has made me able to deliver high-quality projects in a very timely fashion. I am very reliable at my job and will get the work done in time, no matter what. In Addition, I have managed to ensure that the work meets my client's expectations and does not cause an error. I am a hard-working and diligent person who is highly responsible for everything I do. Generally, Freelancing has made me more accountable for doing my job. Additionally, I have had a passion for writing for the last seven years in this field.
0.00
0 Reviews
10+ Question Solved
Related Book For 

The Science And Engineering Of Materials
ISBN: 9781305076761
7th Edition
Authors: Donald R. Askeland, Wendelin J. Wright
Question Posted:
Students also viewed these Engineering questions
-
Determine the eutectoid temperature, the composition of each phase in the eutectoid reaction, and the amount of each phase present in the eutectoid microconstituent for the following systems. For the...
-
Determine the composition of the products of the disassociation reaction CO2 CO + O when the products are at 1 atm and 2500 K.
-
A Nb-60 wt% W alloy is heated to 2800 C. Determine (a) The composition of the solid and liquid phases in both wt% and at%; (b) The amount of each phase in both wt% and at%; and (c) Assuming that the...
-
k) Assume that one of these portfolio's is the Market Portfolio and all portfolios, except Portfolio G, are fairly priced according to the CAPM. What is the highest utility score that can be achieved...
-
Starting from your graphical results of Problem 7b, show graphically the effect on YE and on XM of a depreciation of the nations currency from a position of full employment and a trade deficit.
-
Daisey Company is a very profitable small business. It has not, however, given much consideration to internal control. For example, in an attempt to keep clerical and office expenses to a minimum,...
-
Show that the following eight vectors are pairwise orthogonal: \[\begin{aligned}& s 1=(1,1,0,0,0,0,0,0)^{T} \\& s 2=(0,0,1,1,0,0,0,0)^{T} \\& s 3=(0,0,0,0,1,1,0,0)^{T} \\& s 4=(0,0,,0,0,0,1,1)^{T}...
-
Near the end of its first year of operations, December 31, 2018, Creative Designs Ltd. approached the local bank for a $20,000 loan and was asked to submit financial statements prepared on an accrual...
-
Question C5 [7.5 marks]: The Hastelloy-X plate stock cost from the supplier is $61 /kg and has a material density of 8,220 kg/m3 . The recoverable cost of scrap Hastelloy-X machining chips is $7.8...
-
The adjusted trial balance of Wildwood Company at December 31, 2021, includes the following accounts: D. Wood, Capital $15,600, D. Wood, Drawings $7,000, Service Revenue $39,000, Salaries Expense...
-
Determine the phases present, the compositions of each phase, and the amount of each phase in mol% for the following NiO-MgO ceramics at 2400C: (a) NiO- 30 mol% MgO; (b) NiO-45 mol% MgO; (c) NiO-60...
-
A Bi-60 at% Sb alloy is cooled under equilibrium conditions from the liquid state. Determine (a) The temperature at which the first solid nucleus forms and its composition; (b) The compositions and...
-
Open the data set 2_2_40, which represents the life expectancy of individuals born in the given year for the years 1960 to 2016 in the United States. Construct a time-series plot and comment on any...
-
Research has shown that public speaking ability is a key predictor of career success. Why do you think this is the case?
-
Assume that you work at a bookstore near campus and would like to propose to your supervisor that the bookstore stay open two hours later each evening during the week. You need to choose whether to...
-
Why is it important to consider your audience when analyzing your medium options?
-
Learned that fallacious reasoning can be unethical if you intentionally use it to mislead your audience. What else could you do in an attempt to persuade that could be considered unethical?
-
Some organizations provide the services of mediators to help resolve workplace conflicts. What are the advantages of having a disinterested party resolve a conflict? What are the advantages of having...
-
The current through a 0.5-F capacitor is 6(1-e-t)A. Determine the voltage and power at t=2 s. Assume v(0) = 0.
-
1. Advertising for eyeglasses _________ (increases/decreases) the price of eyeglasses because advertising promotes _________. 2. An advertisement that succeeds in getting consumers to try the product...
-
The codons (words) in DNA (that specify which amino acid should be at a particular point in a protein) are three bases long. How many such three-letter words can be made from the four bases adenine,...
-
Part of a certain DNA sequence is GGTCTATAC. What is the complementary sequence?
-
Which of the noncyclic isomers of C 4 H 7 F are optically active?
-
Use the percentage method to compute the federal income taxes to withhold from the wages or salaries of each employee. Employee Employee Filing Status No. of Withholding No. Name Allowances Gross...
-
Determine the amount to be paid in full settlement of each of two invoices, (a) and (b), assuming that credit for returns and allowances was received prior to payment and that all invoices were paid...
-
Injection Plastics Company has been operating for three years. At December 31, 2023, the accounting records reflected the following: Cash Investments (short-term) $27,500 3,300 Intangibles Accounts...

Study smarter with the SolutionInn App


