Determine the phases present, the compositions of each phase, and the amount of each phase in mol%
Question:
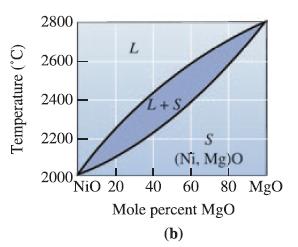
Determine the phases present, the compositions of each phase, and the amount of each phase in mol% for the following NiO-MgO ceramics at 2400°C: (a) NiO- 30 mol% MgO;
(b) NiO-45 mol% MgO;
(c) NiO-60 mol% MgO; and
(d) NiO-85 mol% MgO.
Transcribed Image Text:
Temperature (°C) 2800 2600 2400 2200 L 2000, L+S S (Ni. Mg)0 I NiO 20 40 60 80 MgO Mole percent MgO (b)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (8 reviews)
a The phases are NiO and MgO The ratio of NiO to M...View the full answer

Answered By

Brian Otieno
I'm Brian , an experienced professional freelancer with countless hours of success in freelancing many subjects in different disciplines. Specifically, I have handled many subjects and excelled in many disciplines. I have worked on many Computer Science projects and have been able to achieve a lot in that field. Additionally, I have handled other disciplines like History, Humanities, Social Sciences, Political science, Health care and life science, and Religion / Theology. My experience generally in these subjects has made me able to deliver high-quality projects in a very timely fashion. I am very reliable at my job and will get the work done in time, no matter what. In Addition, I have managed to ensure that the work meets my client's expectations and does not cause an error. I am a hard-working and diligent person who is highly responsible for everything I do. Generally, Freelancing has made me more accountable for doing my job. Additionally, I have had a passion for writing for the last seven years in this field.
0.00
0 Reviews
10+ Question Solved
Related Book For 

The Science And Engineering Of Materials
ISBN: 9781305076761
7th Edition
Authors: Donald R. Askeland, Wendelin J. Wright
Question Posted:
Students also viewed these Engineering questions
-
Determine the phases that are present and the compositions for each phase in Cu-55 wt% Ag at 600C. Data Form Problem 11-23 Consider a Pb-70% Sn alloy. Determine (a) If the alloy is hypoeutectic or...
-
Determine the phases that are present and the compositions for each phase in Cu-85 wt% Ag at 800C. Temperature (C) 1200 1100 1000 900 800 700 600 500 400 300 200 0 10 20 30 40 50 60 70 80 90 100...
-
The unit cell of MgO is shown below. Does MgO have a structure like that of NaCl or ZnS? If the density of MgO is 3.58 g/cm3, estimate the radius (in centimeters) of the O2- anions and the Mg2+...
-
Calculate the following integral approximately by using the Simpson's method: 3.6 f. f(x) dx X 0.0 0.6 1.2 1.8 2.4 3.0 3.6 f(x) 0 0.13 0.05 0.6 0.43 0.74 0.93
-
Identify the advantages of automatic over policy adjustments to correct a trade disequilibrium.
-
The bank portion of the bank reconciliation for Bogalusa Company at October 31, 2019, is shown below. The adjusted cash balance per bank agreed with the cash balance per books at October 31. The...
-
Haar transform (a) For an \(N \times N\) Haar transformation matrix, the Haar basis functions are \[\psi_{k}(t)=\psi_{p q}(t)=\frac{1}{\sqrt{N}} \begin{cases}2^{p / 2}, & (q-1) / 2^{p} \leq t
-
a. From Williss point of view, is litigation or ADR preferred? Why? b. From Telwedas point of view, is litigation or ADR preferred? Why? c. Which form of ADR could be preferred by the two companies?...
-
Image transcription text Task.2b-is-the-capacitor-discharging-below. Current 1 Voltage 0 -1 171 172 173 174 -2 -5 . 50 51 52 53 54 (s) Simulation time (s) Simulation time 0.00 5.6 kS 100 UF HAL 10.0...
-
Pam Corporation acquired all the outstanding stock of Sun Corporation on April 1, 2016, for $15,000,000, when Sun's stockholders' equity consisted of $5,000,000 capital stock and $2,000,000 retained...
-
What is the composition of the Monel alloy?
-
A Nb-W alloy held at 2800C is partly liquid and partly solid. (a) If possible, determine the composition of each phase in the alloy, and (b) If possible, determine the amount of each phase in the...
-
A compound B has the formula C8H10. After vigorous oxidation, it yields benzoic acid (structure in Eq. 17.37). What is the structure of B? Eq. 17.37 Cr(VI) vigorous C OH benzoic acid CH CH Cr(VI)...
-
Your manager asks you to write a type of report not illustrated in this chapter; for example, a compliance report. How would you go about learning the key features of a compliance report and the type...
-
What are five types of evidence you can use in a logical argument?
-
Explain why you should send a thank-you note after all your employment interviews, even with companies you are no longer interested in pursuing.
-
Why is direct organization usually the better choice for business communication?
-
Imagine you have been hired by a global company that is holding a two-week orientation for all new employees at the head office in San Francisco. You will be staying in a hotel for two weeks, and you...
-
In the circuit shown in Fig. 7.81 v(t) = 56e200tV, t > 0 i(t) = 8e200tmA, t > 0 (a) Find the values of R and C. (b) Calculate the time constant Ï. (c) Determine the time required for the voltage...
-
What is an access control list?
-
a. Name each of the following alcohols. b. Name each of the following alcohols, including the stereochemistry if cistrans isomers are possible. HCH,CH,CH,CH,CH3 I CH3 T HCCH2CH 1 CH3 CH3
-
The two isomers having the formula C 2 H 6 O boil at -23 C and 78.5 C. Draw the structure of the isomer that boils at -23 C and of the isomer that boils at 78.5 C.
-
Consider the reaction of propane with chlorine (and appropriate catalyst). a. How many different monochloro products are possible? Name them. b. How many different dichloro products are possible?...
-
Impact on the Balance Sheet Equation. For each transaction, consider the impact on the balance sheet equation (after closing entries are recorded). Thus, if the transactions affect the income...
-
1. The following information pertains to Henry Enterprises for 2023: Cost of goods manufactured Direct labor Direct materials inventory, beginning of year Direct materials inventory, end of year...
-
1. Calculate the missing amount for each of the following notes receivable. Face Value $15,000 Annual Interest Rate Fraction of the Year 4% $25,000 8% $30,000 8 months 4 months 6% 6 months

Study smarter with the SolutionInn App


