A stainless steel is held at 705C under different loads. The following data are obtained: Determine the
Question:
A stainless steel is held at 705°C under different loads. The following data are obtained:
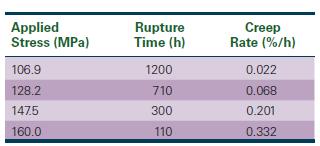
Determine the exponents n and m in Equations 7-22 and 7-23 that describe the dependence of creep rate and rupture time on applied stress.
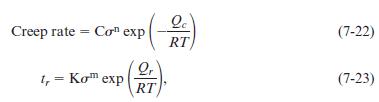
Transcribed Image Text:
Applied Stress (MPa) 106.9 128.2 1475 160.0 Rupture Time (h) 1200 710 300 110 Creep Rate (%/h) 0.022 0.068 0.201 0.332
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (10 reviews)
ANASWER The exponents n and m can be determined using the following equation lnRupt...View the full answer

Answered By

Aketch Cindy Sunday
I am a certified tutor with over two years of experience tutoring . I have a passion for helping students learn and grow, and I firmly believe that every student has the potential to be successful. I have a wide range of experience working with students of all ages and abilities, and I am confident that I can help students succeed in school.
I have experience working with students who have a wide range of abilities. I have also worked with gifted and talented students, and I am familiar with a variety of enrichment and acceleration strategies.
I am a patient and supportive tutor who is dedicated to helping my students reach their full potential. Thank you for your time and consideration.
0.00
0 Reviews
10+ Question Solved
Related Book For 

The Science And Engineering Of Materials
ISBN: 9781305076761
7th Edition
Authors: Donald R. Askeland, Wendelin J. Wright
Question Posted:
Students also viewed these Engineering questions
-
The following data are available for Haul-It-Away Truckers: 1. Compute the predetermined overhead rate for each of the two years, if based on (a) Direct labor hours, (b) Number of moving jobs, and...
-
The following data are for Ernst Company. Note: All inventory is purchased on account, and Accounts Payable relates only to the purchase of inventory. Instructions: Compute the following: 1. The...
-
The following data are taken from the comparative balance sheets prepared for Gubler Wholesale Company. Sales for 2011 were $1,200,000. Sales for 2010 were $1,000,000. 1. Prepare the Assets section...
-
Hehtoc-yrityksen tuloslaskelma on esitetty alla. Yrityksen valmistuskapasiteetti on 120.000 kpl vuodessa. Myyntituotot 80.000 kpl 20 Myytyjen tuotteiden kustannukset Vlittmt aineet Vlitn ty...
-
Explain any duties shareholders assume. Summarize the rights of the shareholders in relationship to other shareholders, the management and directors of the corporation.
-
As a network service provider, XYZ Network Solutions provides high speed network services to many clients such as government, banks, and internet-based businesses. What is the primary security...
-
Extend the analysis of the laminar-flow reactor for a power-law fluid. Perform some computations using the PDEPE solver, and show how the power-law index affects the conversion in the reactor.
-
Aurora Company spent a total of $102,340 in the current period in one of its production centers. In addition, when the production period began, there was work-in-process inventory in the production...
-
In a class of 25 students, 15 were math majors, 17 were computer science majors, and 9 were dual majors in math and computer science. Part: 0 / 4 Part 1 of 4 Use the Venn Diagram tool to illustrate...
-
Homestead Crafts, a distributor of handmade gifts, operates out of owner Emma Finns house. At the end of the current period, Emma reports she has 1,300 units (products) in her basement, 20 of which...
-
What is the difference between failure of a material by creep and that by stress rupture?
-
What is meant by the terms stress rupture and stress corrosion?
-
Arnie's Air Conditioners, Inc., manufactures a variety of heating and cooling equipment. The Super Cool model is one of the higher-demand items and is of particular interest because its manufactured...
-
What is the break-even point for the project? How is it calculated?
-
PMF, Inc. is equally likely to have EBIT this coming year of $7 million, $13 million, or $19 million. Its corporate tax rate is 35%, and investors pay a 15% tax rate on income from equity and a 40%...
-
Describe the three techniques for feasibility analysis.
-
Craxton Engineering will either purchase or lease a new $752,000 fabricator. If purchased, the fabricator will be depreciated on a straight-line basis over seven years. Craxton can lease the...
-
Garnet Corporation is considering issuing risk-free debt or risk-free preferred stock. The tax rate on interest income is 32%, and the tax rate on dividends or capital gains from preferred stock is...
-
Show that a series LR circuit is a lowpass filter if the output is taken across the resistor. Calculate the corner frequency fc if L = 2 mH and R = 10 k .
-
What is the maximum volume of 0.25 M sodium hypochlorite solution (NaOCl, laundry bleach) that can be prepared by dilution of 1.00 L of 0.80 M NaOCl?
-
The vapor pressure of pure benzene is 750.0 torr and the vapor pressure of toluene is 300.0 torr at a certain temperature. You make a solution by pouring some benzene with some toluene. You then...
-
A 4.7 10 -2 mg sample of a protein is dissolved in water to make 0.25 mL of solution. The osmotic pressure of the solution is 0.56 torr at 25 C. What is the molar mass of the protein?
-
The molar mass of a nonelectrolyte is 58.0 g/mol. Determine the boiling point of a solution containing 35.0 g of this compound and 600.0 g of water. The barometric pressure during the experiment was...
-
A partnership has decided to shut down. Total assets amount to $70,000 (all cash). There are no liabilities. Three of the partners have debit balances in their capital accounts of $15,000 each. The...
-
What are the key components of strategic leadership, and how do strategic leaders effectively align organizational goals with external market dynamics and emerging trends to sustain long-term...
-
Describe the role of blogging in business communication today, and explain how to adapt the three-step writing process to blogging and explain some common barriers to successful communication.

Study smarter with the SolutionInn App


