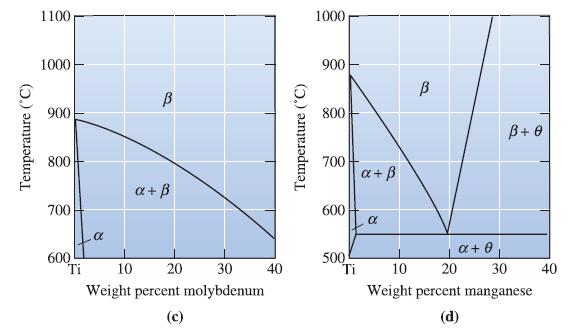
A Ti-Mn alloy is composed of 20 wt% manganese and is kept at 550C. What phase(s) are
Question:
A Ti-Mn alloy is composed of 20 wt% manganese and is kept at 550°C. What phase(s) are present? See Figure 14-8(d).
Transcribed Image Text:
Temperature (°C) 1200 1000 800 600 400 200 Ti В α ‚a + ß 5 10 15 Weight percent tin (a) 20 Temperature (°C) 1200 1100 1000 900 800 700 Ti В 8 a+ß- α + Ti3Al 02 (02₂) 5 10 15 Weight percent aluminum (b) 20
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 87% (8 reviews)
The phases present include i a ferri...View the full answer

Answered By

Brian Otieno
I'm Brian , an experienced professional freelancer with countless hours of success in freelancing many subjects in different disciplines. Specifically, I have handled many subjects and excelled in many disciplines. I have worked on many Computer Science projects and have been able to achieve a lot in that field. Additionally, I have handled other disciplines like History, Humanities, Social Sciences, Political science, Health care and life science, and Religion / Theology. My experience generally in these subjects has made me able to deliver high-quality projects in a very timely fashion. I am very reliable at my job and will get the work done in time, no matter what. In Addition, I have managed to ensure that the work meets my client's expectations and does not cause an error. I am a hard-working and diligent person who is highly responsible for everything I do. Generally, Freelancing has made me more accountable for doing my job. Additionally, I have had a passion for writing for the last seven years in this field.
0.00
0 Reviews
10+ Question Solved
Related Book For 

The Science And Engineering Of Materials
ISBN: 9781305076761
7th Edition
Authors: Donald R. Askeland, Wendelin J. Wright
Question Posted:
Students also viewed these Engineering questions
-
Set up iterated integrals for both orders of integration. Then evaluate the double integral using the easier order and explain why its easier. D is bounded by y = x 3 , y = 2x + 4, x = 0 6x2 dA, D
-
Some hypothetical alloy is composed of 12.5 wt% of metal A and 87.5 wt% of metal B. If the densities of metals A and B are 4.27 and 6.35 g/cm 3 , respectively, whereas their respective atomic weights...
-
Some hypothetical alloy is composed of 12.5 wt% of metal A and 87.5 wt% of metal B. If the densities of metals A and B are 4.27 and 6.35 g/cm3, respectively, whereas their respective atomic weights...
-
Use the Ratio Test to determine the values of x 0 for which each series converges. 00 X 2k 2 k=1 k
-
1. Also the percentage increase for each occupational category. 2. The Excel file Store and Regional Sales Database provides sales data for computers and peripherals showing the store identification...
-
Hendry's Boutique is a retail clothing store for women. The store operates out of a rented building in Storm Lake, Iowa. The following is the store's adjusted year-end trial balance dated December...
-
Table B. 11 presents 38 observations on wine quality. a. Select four observations at random from this data set, then delete these observations and fit a model involving only the regressor flavor and...
-
An engineer decided to make a careful analysis of the cost of fire insurance for his $200,000 home. From a fire rating bureau he found the following risk of fire loss in any year. Outcome Probability...
-
The technology company, Samsung, is considering to revamp an old tablet product of theirs and would like for it to make a big impression against other competitors in this day and age. -What branding...
-
The Web 2.0 revolution has changed the way businesses communicate with consumers and within their own organizations. Podcasts, wikis, and blogs can be used to build reputations, communicate with...
-
A 5182-O aluminum alloy part that had been exposed to salt water showed severe corrosion along the grain boundaries. Explain this observation based on the expected phases at room temperature in this...
-
Would you expect a 2024-T9 aluminum alloy to be stronger or weaker than a 2024-T6 alloy? Explain.
-
In Exercises, determine the adjacency matrix of the given digraph. a. b. c. VI V3 VA V2 VA VI ys N3 VA
-
Identify three ways that capital is transferred between savers and borrowers.
-
Explain how the cash flows are structured in order to estimate the net advantage to leasing.
-
What are the main benefits of going private?
-
What is a private equity fund?
-
Describe the process of securitization as applied to home mortgages.
-
Metabolism of arginine produces urea and the rare amino acid ornithine. Ornithine has an isoelectric point close to 10. Propose a structure for ornithine.
-
For the given transfer function: Vo(s) / Vi(s) = (s^2C^2R^2 + 1) / (s^2C^2R^2 + 4sCR + 1) Assumiing that 1/(CR) = 120 PI so write the matlab code to find the magnitude plot
-
Use the matrix inverse and the matrix division method to solve the following set for x and y in terms of c: 4cx + 5y = 43 3x 4y = -22
-
The currents i 1 , i 2 , and i 3 in the circuit shown in Figure P12 are described by the following equation set if all the resistances are equal to R. Here 1 and 2 are applied voltages; the other...
-
The equations for the armature-controlled dc motor shown in Figure P13 follow. The motors current is i, and its rotational velocity is 2. where L, R, and I are the motors inductance, resistance, and...
-
A government agency in charge of transportation has completed emergency project work three days earlier than the target they previously communicated. What is the best method to communicate this...
-
A 50-year-old man is admitted for a femur fracture following a motor vehicle collision. Two days after admission, he becomes very agitated, tremulous, diaphoretic, tachycardic, and hypertensive. From...
-
Company X has been experiencing high employee turnover rates in recent years. As a newly hired HR manager, what steps would you take to address this issue and improve employee retention? Identify and...

Study smarter with the SolutionInn App


