An electrochemical cell consists of a silver metal electrode immersed in a solution with [Ag + ]
Question:
An electrochemical cell consists of a silver metal electrode immersed in a solution with [Ag+] = 1.0 M separated by a porous disk from a copper metal electrode. If the copper electrode is placed in a solution of 5.0 M NH3 that is also 0.010 M in Cu(NH3)42+, what is the cell potential at 25οC?
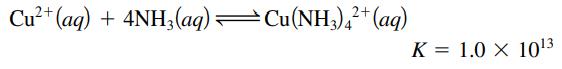
Transcribed Image Text:
Cu²+ (aq) + 4NH3(aq) —Cu(NH3)4²+ (aq) K = 1.0 X 10¹3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (8 reviews)
The cell potential at 25C Ecell Ag10 M 00060 M Cu2aq50 M0010 ...View the full answer

Answered By

Brian Otieno
I'm Brian , an experienced professional freelancer with countless hours of success in freelancing many subjects in different disciplines. Specifically, I have handled many subjects and excelled in many disciplines. I have worked on many Computer Science projects and have been able to achieve a lot in that field. Additionally, I have handled other disciplines like History, Humanities, Social Sciences, Political science, Health care and life science, and Religion / Theology. My experience generally in these subjects has made me able to deliver high-quality projects in a very timely fashion. I am very reliable at my job and will get the work done in time, no matter what. In Addition, I have managed to ensure that the work meets my client's expectations and does not cause an error. I am a hard-working and diligent person who is highly responsible for everything I do. Generally, Freelancing has made me more accountable for doing my job. Additionally, I have had a passion for writing for the last seven years in this field.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Chemistry
ISBN: 9781305957404
10th Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Question Posted:
Students also viewed these Engineering questions
-
An electrochemical cell consists of a silver metal electrode immersed in a solution with [Ag + ] = 1.00 M separated by a porous disk from a compartment with a copper metal electrode immersed in a...
-
An electrochemical cell consists of a nickel metal electrode immersed in a solution with [Ni 2+ ] = 1.0 M separated by a porous disk from an aluminum metal electrode. a. What is the potential of this...
-
Gooran, Inc., has current assets of $240 million; property, plant, and equipment of $380 million; and other assets totaling $120 million. Current liabilities are $170 million and long-term...
-
How could a system be designed to allow a choice of operating systems from which to boot? What would the bootstrap program need to do?
-
Wii Brothers, a game manufacturer, has a new idea for an adventure game. It can market the game either as a traditional board game or as an interactive DVD, but not both. Consider the following cash...
-
Explain the relationship between climate change and food choice.
-
Melodic Musical Sales, Inc. is located at 5500 Fourth Avenue, City, ST 98765. The corporation uses the calendar year and accrual basis for both book and tax purposes. It is engaged in the sale of...
-
The picture shows six different situations of a box with one or two horizontal forces applied. The mass of each box is given, and the strength of each force is specified in terms of F. In each case...
-
Post the adjusting entries to the ledger accounts. Enter the totals from the trial balance as beginning account balances. (Use T-Accounts.) (Post entries in the order of journal entries presented in...
-
Cadmium sulfide is used in some semiconductor applications. Calculate the value of the solubility product constant (K sp ) for CdS given the following standard reduction potentials: CdS (s) + 2e ...
-
Consider a concentration cell that has both electrodes made of some metal M. Solution A in one compartment of the cell contains 1.0 M M 2+ . Solution B in the other cell compartment has a volume of...
-
The Kellogg Bank requires borrowers to keep an 8 percent compensating balance. Gorman Jewels borrows $340,000 at a 7 percent stated APR. What is the effective interest rate on the loan?
-
Using appropriate graphs, consider the implications, for both a perfectly competitive market and a monopoly, if the marginal cost of production falls. Given your conclusions, would it always be true...
-
Bridgeport Windows Inc is in the process of setting a target price on its newly designed tinted window. Cost data relating to the window at a budgeted volume of 3,440 units are as follows. Per Unit...
-
How do geopolitical dynamics influence the trajectory of market trends within technologically advanced sectors ?
-
A government faced with excessive inflation in the economy could attempt to reverse the situation by taking what steps ?
-
What The spending approach (demand) for measuring gross domestic product (GDP) adds together?
-
What mass of water at 25.0C added to a Styrofoam cup containing two 50.0-g ice cubes from a freezer at 15.0C will result in a final temperature of 5.0C for the drink?
-
Differentiate. y = ln(3x + 1) ln(5x + 1)
-
A bowling ball has a weight of 12 lb and the length of the lane is approximately 60. feet. Treat the ball in the lane as a one-dimensional box. What quantum number corresponds to a velocity of 7.5...
-
For a particle in a two-dimensional box, the total energy eigenfunctions are a. Obtain an expression for E nx , n y in terms of n x , n y , a, and b by substituting this wave function into the two...
-
Consider the contour plots of Problem P15.17. a. What are the most likely area or areas Îx Îy to find the particle for each of the eigenfunctions of HË depicted in plots af? b. For...
-
A pitcher throws a 0.60 kg ball of clay at a 4.0 kg block of wood. The clay sticks to the wood on impact, and their joint velocity afterward is 2.5 m/s. What was the original speed of the clay (in...
-
A helium-filled balloon, whose envelope has a mass of 0.29 kg, is tied to a 3.8-m long, 0.054-kg string. The balloon is spherical with a radius of 0.40 m. When released, it lifts a length h of the...
-
Give a summary of concepts and the applications for the following quistion: When a 200.0-g mass is attached to a spring, it stretches the spring by 7.50 cm. With that mass-spring system in...

Study smarter with the SolutionInn App


