Cadmium sulfide is used in some semiconductor applications. Calculate the value of the solubility product constant (K
Question:
Cadmium sulfide is used in some semiconductor applications. Calculate the value of the solubility product constant (Ksp) for CdS given the following standard reduction potentials:
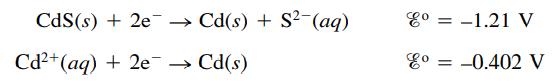
Transcribed Image Text:
CdS (s) + 2e →→ Cd(s) + S² (aq) Cd²+ (aq) + 2e → Cd(s) ४० = -1.21 V co = -0.402 V
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (9 reviews)
Thorough solution with explanation The solubility product ...View the full answer

Answered By

Labindao Antoque
I graduated in 2018 with a Bachelor of Science degree in Psychology from Dalubhasaan ng Lungsod ng San Pablo. I tutored students in classes and out of classes. I use a variety of strategies to tutor students that include: lecture, discussions about the subject matter, problem solving examples using the principles of the subject matter being discussed in class , homework assignments that are directed towards reinforcing what we learn in class , and detailed practice problems help students to master a concept. I also do thorough research on Internet resources or textbooks so that I know what students need to learn in order to master what is being taught in class .
0.00
0 Reviews
10+ Question Solved
Related Book For 

Chemistry
ISBN: 9781305957404
10th Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Question Posted:
Students also viewed these Engineering questions
-
For the circuit in Fig. 6.70, calculate the value of R that will make the energy stored in the capacitor the same as that stored in the inductor under dc conditions. 160 uF 5A 2 4 mH
-
Calculate the value of the consumption function at each level of disposable income in Table 1 if a = 100 and mpc = 09.
-
Calculate the value of a $1,000 bond which has 10 years until maturity and pays quarterly interest at an annual coupon rate of 12 percent. The required return on similar-risk bonds is 20 percent.
-
The comparative balance sheet of Merrick Equipment Co. for December 31, 20Y9 and 20Y8, is as follows: Dec. 31, 20Y9 Dec. 31, 20Y8 Assets Cash $70,720 $47,940 Accounts receivable (net) 207,230 188,190...
-
Suppose you are a depositor at Melvin's Bank, which has the balance sheet shown in Table 10.1A. Deposit insurance does not exist. You originally deposited your money in Melvin's Bank because its...
-
Diesel fuel (C12H26) at 25C is burned in a steady flow combustion chamber with 20 percent excess air that also enters at 25C. The products leave the combustion chamber at 500 K. Assuming combustion...
-
The front door of a dishwasher of width \(580 \mathrm{~mm}\) has a vertical air vent that is \(500 \mathrm{~mm}\) in height with a \(20-\mathrm{mm}\) spacing between the inner tub operating at...
-
Assume that the risk-free rate is 6% and that the expected return on the market is 13%. What is the required rate of return on a stock that has a beta of 0.7?
-
Occasionally, someone claims to have built a perpetual motion machine that can generate energy indefinitely from nothing. For this discussion topic, address the following. Why are perpetual motion...
-
Ohio Logistics manages the logistical activities for firms by matching companies that need products shipped with carriers that can provide the best rates and best service for the companies. Ohio...
-
The electrolysis of BiO + produces pure bismuth. How long would it take to produce 10.0 g Bi by the electrolysis of a BiO + solution using a current of 25.0 A?
-
An electrochemical cell consists of a silver metal electrode immersed in a solution with [Ag + ] = 1.0 M separated by a porous disk from a copper metal electrode. If the copper electrode is placed in...
-
Raj Cycles Limited is a leading bespoke cycle retailer and it operates throughout the year. The cost of goods sold last year was 835,000. The firm modifies eight items to customize cycles as per...
-
S Last month when Holiday Creations, Incorporated, sold 37,000 units, total sales were $148,000, total variable expenses were $111,000, and fixed expenses were $36,500. Required: 1. What is the...
-
Example: A high-tech consumer product manufacturer in Southeast Wisconsin (Milwaukee) is investigating outsourcing possibilities for an important electronic component used in their final product. The...
-
JP Morgan Bank classifies its investments into the following categories: (a) Trading bond investments. (b) Available-for-Sale bond investments, (c) Held-to-Maturity bond investments (d) Equity...
-
Complete a tax return for Stewart L. Benjamin. Stewart is unmarried, has no dependents, and works as a retail manager; his W-2 is below. Using the1099s and the transaction description which follow,...
-
Finch Manufacturing Company established the following standard price and cost data: Sales price Variable manufacturing cost Fixed manufacturing cost Fixed selling and administrative cost $ 8.20 per...
-
In Exercise draw a graph with the given adjacency matrix. 1010 1101 0011 0011
-
Differentiate. y = ln(3x + 1) ln(5x + 1)
-
Reaction of iodoethane with CN yields a small amount of isonitrile, CH3CH2N C, along with the nitrile CH3CH2C N as the major product. Write electron-dot structures for both products, assign formal...
-
A Alkynes can be made by dehydrohalogenation of vinylic halides in a reaction that is essentially an E2 process. In studying the stereochemistry of this elimination, it was found that...
-
(S)-2-Butanol slowly racemizes on standing in dilute sulfuric acid.Explain. CH3CH2CHCH3 2-Butanol
-
Describe, in steps, at least one strategy used to attack embedded operating systems. Describe, in steps, at least one strategy used to defend against the chosen attack.
-
how does a hypercompetetice environment challenge those in an industry
-
Describe how managers can reduce unnecessary costs, including real - world examples of how it has been done

Study smarter with the SolutionInn App


