At a particular temperature, 8.1 moles of NO 2 gas are placed in a 3.0-L container. Over
Question:
At a particular temperature, 8.1 moles of NO2 gas are placed in a 3.0-L container. Over time the NO2 decomposes to NO and O2:
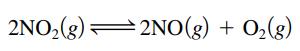
At equilibrium the concentration of NO(g) was found to be 1.4 mol/L. Calculate the value of K for this reaction.
Transcribed Image Text:
2NO₂(g) 2NO(g) + O₂(g)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 70% (10 reviews)
The value of K ra...View the full answer

Answered By

Willis Omondi
Hi, I'm Willis Omondi, a proficient and professional academic writer. I have been providing high-quality content that best suits my clients and completing their work within the deadline. All my work has been 100% plagiarism-free, according to research from my services, especially in arts subjects and many others
0.00
0 Reviews
10+ Question Solved
Related Book For 

Chemistry
ISBN: 9781305957404
10th Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Question Posted:
Students also viewed these Engineering questions
-
Two moles of an ideal gas are placed in a container whose volume is 8.5 10-3 m3. The absolute pressure of the gas is 4.5 105 Pa. What is the average translational kinetic energy of a molecule of...
-
At a particular temperature and pressure, a helium gas contains 5 10 25 atoms/m 3 . If a 10-kV/m field applied to the gas causes an average electron cloud shift of 10 8 m, find the dielectric...
-
At a particular temperature the value of [Ba 2+ ] in a saturated solution of barium sulfate is 1.035x10 -5 M . Starting with this information, calculate the K sp of barium sulfate at this temperature.
-
State whether the following statement are True or False An increase in the owner(s) equity, in the absence of any further investment by the owners is typically effected by sale transactions.
-
Explain the functions of distribution channels.
-
You are the controller of Classic Toys Ltd. Kathy McDonnell, the president, recently mentioned to you that she found an error in the 2019 financial statements, which she believes has corrected...
-
When applying a classical variables sampling plan, such as ratio or difference estimation or MPU estimation, an auditor must estimate the risk of incorrect rejection, the risk of incorrect...
-
Chino Company manufactures fabric and clothing. Managers can either sell the unfinished fabric to other clothing manufacturers or incur additional conversion costs to create a finished garment. The...
-
Part One: Please summarize the five steps required for Metallic Cartridge Reloading. Part Two: What safety precautions are needed as part of the process? Part Three: Finally, describe the best...
-
A maker of energy drinks is considering abandoning can containers and going exclusively to bottles because the sales manager believes customers prefer drinking from bottles. However, the vice...
-
A sample of solid ammonium chloride was placed in an evacuated chamber and then heated, causing it to decompose according to the following reaction: In a particular experiment, the equilibrium...
-
Methanol, a common laboratory solvent, poses a threat of blindness or death if consumed in sufficient amounts. Once in the body, the substance is oxidized to produce formaldehyde (embalming fluid)...
-
Calculate and compare the risk (betas) of the following investments: (a) A share of Apple stock; (b) A one-year call option on Apple; (c) A one-year put option; (d) A portfolio consisting of a share...
-
Do the borrowers have sufficient funds for closing and reserves? Nontraditional credit sources Verification of mortgage Verification of deposit Did the borrowers make their housing payments on time?...
-
Assuming the Reddings refinance their home and use the proceeds to retire debt, what is the tax consequence in the current tax year if one point is paid to secure the interest rate?
-
You have been ineligible for financial aid since Fall 2021. Please outline what impeded your progress during those terms and how those issues have been resolved?
-
Video: Bonds are 500 years old and bigger than ever https://www.bloomberg.com/news/videos/2017-10-02/bonds-are-500-years-old-and-bigger-than-ever-video. What are your thoughts about this video?
-
Michael Wittry has been investing in his Roth IRA retirement account for 18 years. Three years ago, his account was worth $215,672. After losing 1/4 of its original value, it then gained 1/3 of its...
-
Section 29.8 states that the total energy released by the proton-proton cycle is the same as that released by the carbon cycle. Why must the total energy released be the same?
-
One Way Cellular accountants have assembled the following data for the year ended September 30, 2014: Prepare the operating activities section using the indirect method for One Way Cellulars...
-
For each of the following compounds, predict the energy barrier to rotation (looking down any one of the C-C bonds). Draw a Newman projection and then compare the staggered and eclipsed...
-
In each case below, identify the highest and lowest energy conformations. In cases where two or three conformations are degenerate, draw only one as your answer. (a) (b) (c) (d)
-
Compare the three staggered conformations of ethylene glycol. The anti conformation of ethylene glycol is not the lowest energy conformation. The other two, staggered conformations are actually lower...
-
f(x) = x In(2x) Find f(x) for the function below....
-
Explain the importance of leadership to a team. Explain leadership practices you incorporated into your leadership style. Provide examples of how you incorporated those leadership practices within...
-
The severity of many sports injuries is related to the magnitude of the acceleration that an athlete's body undergoes as it comes to rest, especially when joints (such as ankles and knees) are not...

Study smarter with the SolutionInn App


