Balance the following equations representing combustion reactions: a. b. (0) + (g) OH (g) + C 90
Question:
Balance the following equations representing combustion reactions:
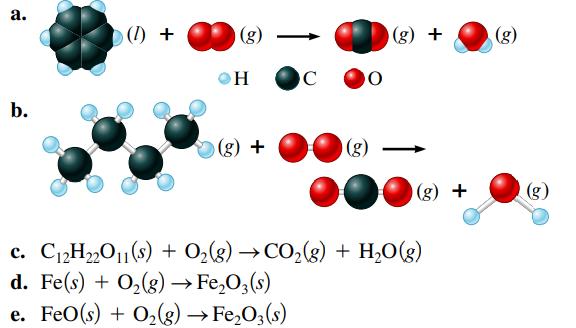
Transcribed Image Text:
a. b. (0) + (g) OH (g) + C 90 (g) (g) + (g) + c. C₁2H₂2O11(s) + O₂(g) →CO₂(g) + H₂O(g) d. Fe(s) + O₂(g) → Fe₂O3(s) e. FeO(s) + O₂(g) → Fe₂O3(s) (g) (g)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 72% (11 reviews)
ANSWER Balancing a chemical equation involves adjusting the coefficients numbers in front of each mo...View the full answer

Answered By

Dennis Nyangau
I have been tutoring for several years now, and I absolutely love it! I love being able to help students one-on-one and see them succeed. It is so gratifying to see a student understand a concept that they were struggling with before. I also enjoy getting to know my students and helping them to reach their full potential.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Chemistry
ISBN: 9781305957404
10th Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Question Posted:
Students also viewed these Engineering questions
-
Balance the following equations and indicate whether they are combination, decomposition, or combustion reactions: (a) C3H6(g) + O2 (g) CO2 (g) + H2O(g) (b) NH4NO3(s) N2O(g) + H2O(g) (c) C5H6O(I) +...
-
Balance the following equations and indicate whether they are combination, decomposition, or combustion reactions: (a) PbCO3(s) PbO(s) + CO2(g) (b) C2H4(g) + O2(g) CO2(g) + H2O(g) (c) Mg(s) + N2(g)...
-
Balance the following equations and write the corresponding ionic and net ionic equations (if appropriate): (a) (b) (c) Ba(OH)-(aq) + HPO 4 (aq )- HCIO4 (aq) + Mg(OH )2 (s)
-
Show that 15 is an inverse of 7 modulo 26.
-
Dan and Diana file a joint return. Dan earned $31,000 during the year before losing his job. Diana received Social Security benefits of $5,000. a. Determine the taxable portion of the Social Security...
-
An observer is 20 m above the ground floor of a large hotel atrium looking at a glass-enclosed elevator shaft that is 20 m horizontally from the observer (see figure). The angle of elevation of the...
-
What approaches to controlling COVID-19 succeeded, and what approaches were ineffective? In the United States, the effects of COVID-19 exposed racial and ethnic inequalities and, in some cases,...
-
The TransCanada Lumber Company and Mill processes 10,000 logs annually, operating 250 days per year. Immediately upon receiving an order, the logging companys supplier begins delivery to the lumber...
-
Discuss the role of thread scheduling in a multi-threaded environment. What factors determine thread priority, and how do operating systems ensure fair CPU time allocation among competing threads ?
-
Prove that each of the following formulas for (v. tv) defines an inner product on R3. Verify all the inner product axioms in careful detail: (a) V1W1 + 2V2w2 + 3v3w3 (b) 4v1w1 + 2 v1w2 + 2v2wi +4v2W2...
-
Glass is a mixture of several compounds, but a major constituent of most glass is calcium silicate, CaSiO 3 . Glass can be etched by treatment with hydrofluoric acid; HF attacks the calcium silicate...
-
Balance each of the following chemical equations. a. KO (s) + HO(1)KOH(aq) + O(g) + HO(aq) b. FeO3(s) + HNO3(aq) Fe(NO3)3(aq) + HO(1) c. NH3(g) + O(g) NO(g) + HO(g) d. PC1,(1) + HO(1) H3PO4(aq) +...
-
What are the measurement issues to consider when using return on investment (ROI)?
-
Analyse the scenario and discuss whether a blockchain solution would be appropriate for this domain. You must refer to one or more of the Blockchain Suitability flowcharts You cant just simply answer...
-
How much tax will Hanna pay on her qualified dividends and capital gains?
-
The finance controller is seeking process improvement to reduce customer complaints due to late billings. The concerned departments include customer service who receives and processes orders,...
-
January Taxes February Taxes March Taxes Q 1 Earnings FWT $ 1 2 , 4 0 0 $ 1 2 , 7 2 0 $ 1 2 , 1 2 0 $ 3 3 1 , 2 0 0 Social Security $ 7 , 5 7 9 . 5 0 $ 7 , 2 4 0 . 3 6 $ 7 , 9 9 0 . 1 3 $ 3 6 7 , 9 0...
-
1. A note made on January 4 and due in 90 days would mature on what date? (assume February has 28 days) a. April 2 c. April 4 b. April 3 d. April 5 2. The maturity value of a $5,000, 60-day, 6...
-
Using the semi-annually compounded yield curve in Table 2.4, price the following securities: (a) 5-year zero coupon bond (b) 7-year coupon bond paying 15% semiannually (c) 4-year coupon bond paying...
-
Use the method of Example 4.29 to compute the indicated power of the matrix. 1 0 1
-
Calculate the viscosity of benzene vapour at (a) 273 K, (b) 298 K, (c) 1000 K. Take a~ 0.88 nM-1.
-
Calculate the thermal conductivities of (a) Neon, (b) Nitrogen at 300 K and 15 mbar. Each gas is confined in a cubic vessel of side 15 cm, one wall being at 305 K and the one opposite at 295 K. What...
-
The viscosity of a chlorofluorocarbon (CFC) was measured by comparing its rate of flow through a long narrow tube (using Poiseuille formula) with that of argon. For the same pressure differential,...
-
1. A spotlight on a boat pictured below is 2.5 m above the water. A spotlight strikes the water at a point that is 8 meters behind the boat on the water's surface. The depth of the water is 4 m. How...
-
1. A high school athlete runs 1.00 x 102 m in 12.20 s. What is the velocity in m/s and km/h? 2. A person walks. 13 km in 2.0 h. What is the person's average velocity in km/h and m/s? 5. Suppose a car...
-
Suppose that a 0.225 m radius, 500 turn coil produces an average emf of 8800 V when rotated one-fourth of a revolution in 4.08 ms, starting from its plane being perpendicular to the magnetic field....

Study smarter with the SolutionInn App


