Balance each of the following chemical equations. a. KO (s) + HO(1)KOH(aq) + O(g) + HO(aq) b.
Question:
Balance each of the following chemical equations.
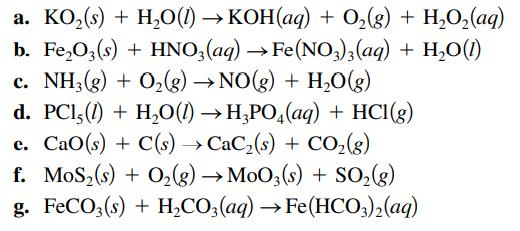
Transcribed Image Text:
a. KO₂ (s) + H₂O(1)→KOH(aq) + O₂(g) + H₂O₂(aq) b. Fe₂O3(s) + HNO3(aq) → Fe(NO3)3(aq) + H₂O(1) c. NH3(g) + O₂(g) → NO(g) + H₂O(g) d. PC1,(1) + H₂O(1)→ H3PO4(aq) + HCl(g) e. CaO(s) + C(s) →CaCz(s) + CO,(g) f. MoS₂ (s) + O₂(g) → MoO3(s) + SO₂(g) g. FeCO3(s) + H₂CO3(aq) →Fe(HCO3)2(aq)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 41% (12 reviews)
2KO2s 2H2Ol 4KOHaq O2g 2H2O2aq Fe2O3s 6...View the full answer

Answered By

Tobias sifuna
I am an individual who possesses a unique set of skills and qualities that make me well-suited for content and academic writing. I have a strong writing ability, allowing me to communicate ideas and arguments in a clear, concise, and effective manner. My writing is backed by extensive research skills, enabling me to gather information from credible sources to support my arguments. I also have critical thinking skills, which allow me to analyze information, draw informed conclusions, and present my arguments in a logical and convincing manner. Additionally, I have an eye for detail and the ability to carefully proofread my work, ensuring that it is free of errors and that all sources are properly cited. Time management skills are another key strength that allow me to meet deadlines and prioritize tasks effectively. Communication skills, including the ability to collaborate with others, including editors, peer reviewers, and subject matter experts, are also important qualities that I have. I am also adaptable, capable of writing on a variety of topics and adjusting my writing style and tone to meet the needs of different audiences and projects. Lastly, I am driven by a passion for writing, which continually drives me to improve my skills and produce high-quality work.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Chemistry
ISBN: 9781305957404
10th Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Question Posted:
Students also viewed these Engineering questions
-
Balance each of the following chemical equations. a. KO2(s) + H2O(l) ( KOH(aq) + O2(g) + H2O2(aq) b. Fe2O3(s) + HNO3(aq) ( Fe(NO3)3(aq) + H2O(l) c. NH3(g) + O2(g) ( NO(g) + H2O(g) d. PCl5(l) + H2O(l)...
-
Balance each of the following oxidation reduction reactions by using the oxidation states method. a. C2H6(g) + O2(g) CO2(g) + H2O(g) b. Mg(s) + HCl(aq) Mg2+(aq) + Cl2(aq) + H2(g) c. Cu(s) + Ag+(aq)...
-
Each of the following equations describes a reaction of a compound called methyl formate. To what class of compounds does methyl formate belong? Which reactions require a reducing agent? Which...
-
IfAUB=AUC and An B=An C, then B = C. Statement-2 AU (BOC) = (AUB) n (AUC)
-
Beth retires when she turns 65. She begins receiving a monthly pension of $300 from her employers qualified retirement plan. While employed, Beth contributed $13,000 to the plan. a. Beth uses the...
-
A camera is set up at the starting line of a drag race 50 ft from a dragster at the starting line (camera 1 in the figure). Two seconds after the start of the race, the dragster has traveled 100 ft...
-
How did the COVID-19 crisis affect you personally? In the United States, the effects of COVID-19 exposed racial and ethnic inequalities and, in some cases, exacerbated tensions. In the spring of...
-
Queensland Enterprises needs a cash budget for the month of June. The following information is available: The cash balance on June 1 is $13,000. Sales for May and June are $40,000 and $50,000,...
-
Morganton Company makes one product and it provided the following information to help prepare the master budget: a. The budgeted selling price per unit is $65. Budgeted unit sales for June, July,...
-
Wedding Planners Limited (WP), owned by Anne and Francois Tremblay, provides wedding planning and related services. WP owns a building (the Pavilion) that has been custom-made for hosting weddings....
-
Balance the following equations representing combustion reactions: a. b. (0) + (g) OH (g) + C 90 (g) (g) + (g) + c. C2H2O11(s) + O(g) CO(g) + HO(g) d. Fe(s) + O(g) FeO3(s) e. FeO(s) + O(g) FeO3(s)...
-
Iron oxide ores, commonly a mixture of FeO and Fe 2 O 3 , are given the general formula Fe 3 O 4 . They yield elemental iron when heated to a very high temperature with either carbon monoxide or...
-
1. What are the ethical issues in this case from an employees point of view? From managements point of view? From a consumers point of view? 2. Who are the stakeholders , and what are their stakes?...
-
The objective of the assignment is exposing the students to new sustainable technologies ( products , processes , software, etc. ) and the Life Cycle Analysis of Such technologies. The students will...
-
The following numbers apply to Rockfire Grill for 2 0 2 1 Net income before tax = $ 8 6 , 0 0 0 Unrealized gain on available - for - sale securities before tax = $ 7 3 , 0 0 0 Loss from foreign...
-
Iron Chemicals acquires a machine that should go through a major overhaul every three years. The total price for the equipment is 1 million. It is estimated that each overhaul will cost 200,000. The...
-
For each of the following situations, indicate the effect on assets, net income, and retained earnings at December 3 1 , 2 0 2 0 and 2 0 2 1 as follows: O if the financial statement element is...
-
Introduction: Supply chain management (SCM) plays a critical role in the success and competitiveness of businesses across various industries. Auditing SCM processes for efficiency and transparency is...
-
Compute the price, the yield and the continuously compounded yield for the following Treasury bills. For the 1-year Treasury bill also compute the semi-annually compounded yield. (a) 4-week with...
-
You are standing at x = 9.0 km and your assistant is standing at x = 3.0 km. Lightning bolt 1 strikes at x = 0 km and lightning bolt 2 strikes at x = 12.0 km. You see the flash from bolt 2 at t = 10...
-
Calculate the thermal conductivity of nitrogen (C; m = 20.8 J K-1 mol-1, a= 0.43 nm3) at room temperature (20C).
-
Calculate the diffusion constant of nitrogen at 25C and (a) 10,0 Pa, (b) 100 kPa, (c) 15.0 MPa. If a pressure gradient 01'0.20 bar m-1 is established in a pipe, what is the flow of gas due to...
-
The mobility of an acetate ion in aqueous solution at 25C is 4.24 x 10-8 m3 S-1 V-1. Calculate the molar ionic conductivity.
-
Consider the general case of a block of mass MB hanging at rest and a dart of mass mo moving horizontally with speed vo. The dart hits and embeds in the block, and the center of mass of the...
-
3 kg of water, originally at 10 degrees Celsius, is mixed with 10 kg of lead. The final result at equilibrium is 16 degrees Celsius. Find the initial temperature of the lead. C = 4186 kg-C (specific...
-
Write a java program to perform to find out area of a square?

Study smarter with the SolutionInn App


