Balance the following oxidationreduction reactions that occur in acidic solution using the half-reaction method. a. Cu(s) +
Question:
Balance the following oxidation–reduction reactions that occur in acidic solution using the half-reaction method.
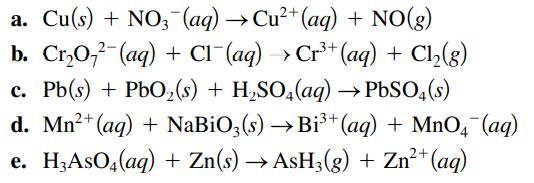
Transcribed Image Text:
a. Cu(s) + NO3(aq) → Cu²+ (aq) + NO(g) b. Cr₂O72 (aq) + Cl¯(aq) → Cr³+ (aq) + Cl₂(g) c. Pb(s) + PbO₂ (s) + H₂SO4(aq) → PbSO4(s) d. Mn²+ (aq) + NaBiO3(s) →Bi³+ (aq) + MnO4 (aq) e. H,AsO4(aq) + Zn(s) → AsH3(g) + Zn²+ (aq)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 37% (8 reviews)
a Cus NO3aq Cuaq NOg Halfreaction Oxidation Cus Cuaq Reduction NO3aq N...View the full answer

Answered By

Hande Dereli
Enthusiastic tutor, skilled in ACT and SAT tutoring. Raised one student's score on the SATs from 1100 combined to 1400. Graduated with a 3.9 GPA from Davidson College and led a popular peer tutoring group for three years. Scored in the top 0.06% in the nation on the SATs. The real reason I'm the one to help you nail the test? Results. Clients invariably praise my ability to listen and communicate in a low-stress, fun way. I think it's that great interaction that lets me raise retest SAT scores an average of 300 points.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Chemistry
ISBN: 9781305957404
10th Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Question Posted:
Students also viewed these Engineering questions
-
Balance each of the following oxidation reduction reactions by using the oxidation states method. a. C2H6(g) + O2(g) CO2(g) + H2O(g) b. Mg(s) + HCl(aq) Mg2+(aq) + Cl2(aq) + H2(g) c. Cu(s) + Ag+(aq)...
-
Balance these redox reactions that occur in aqueous solution. Use whatever water-derived species is necessary; there may be more than one correct balanced equation. a. CrO3 + Ni2+ ( Cr3+ + Ni3+ b....
-
Balance these redox reactions that occur in aqueous solution. Use whatever water-derived species is necessary; there may be more than one correct balanced equation. a. ClO + Ti2+ ( Ti4+ + Cl b. BrO3...
-
My division had another great year last year. We all worked hard, and the results were there. But again we got no reward for our hard work. It's very frustrating. - Division Manager, General Products...
-
What problem may exist in determining the amount realized for an investor who exchanges common stock of a publicly traded corporation for a used building? How is the problem likely to be resolved?
-
What physical properties are used to identify minerals?
-
How is proving manhood related to gender ideology, and how is it expressed in society?
-
An excerpt from the Sun Company's annual report is presented below. Calculating any profit measures deemed necessary, discuss the implications of the profitability of thecompany. Sun Company Income...
-
The Devon Motor Company produces automobiles. On April 1, the company had no beginning inventories, and it purchased 6,290 batteries at a cost of $95 per battery. It withdrew 5,800 batteries from the...
-
Bob and carol was divorced and Bob no longer wanted to keep the $100,000 life insurance policy he had previously purchased. He sold the policy to his friend Bill for $10,000 and Bill named himself...
-
Specify which of the following equations represent oxidation reduction reactions, and indicate the oxidizing agent, the reducing agent, the species being oxidized, and the species being reduced. a....
-
A 30.0-mL sample of an unknown strong base is neutralized after the addition of 12.0 mL of a 0.150 M HNO 3 solution. If the unknown base concentration is 0.0300 M, give some possible identities for...
-
Assume that the Poisson distribution has the indicated mean and use Formula 4-10 to find the probability of the value given for the random variable x. = 0.250, x = 2
-
Effective May 1 , the shareholders of Chiefs Corporation approved a 2 - for - 1 stock split of the companys common stock. Prior to the stock split, Chiefs shareholder s equity items were as follows:...
-
The Great Indoors sells carpet and floor coverings. The company will pay stockholders a $0.95 quarterly dividend later this afternoon . Investors anticipate that the company will increase this...
-
On January 2 of the current year, BC Co. acquired 1,000 shares of its $10 par value common stock for $20,000. On July 1, BC exchanged this stock for land to be held for use as the site of the...
-
The Fancy Umbrella Company makes beach umbrellas. The production process requires 3 square meters of plastic sheeting and a metal pole. The plastic sheeting costs $0.50 per square meter and each...
-
What are the correct Journal Entries for these 2 transactions. Decided to sell second line of consulting business. Found a buyer for second line of consulting services. Sold the business in exchange...
-
The accounts in the ledger of Hickory Furniture Company as of December 31, 2016, are listed in alphabetical order as follows. All accounts have normal balances. The balance of the cash account has...
-
What kind of rays are X-rays?
-
A copper penny can be dissolved in nitric acid but not in hydro chloric acid. Using reduction potentials from the book, show why this is so. What are the products of the reaction? Newer pennies...
-
You want to plate out nickel metal from a nickel nitrate solution onto a piece of metal inserted into the solution. Should you use copper or zinc? Explain.
-
Galvanic cells harness spontaneous oxidationreduction reactions to produce work by producing a current. They do so by controlling the flow of electrons from the species oxidized to the species...
-
The following data refers to Huron Corporation for the year 20x2. Sales revenue $ 2,106,000 Raw-material inventory, 12/31/x1 89,000 Purchases of raw material in 20x2 731,000 Raw-material inventory,...
-
As Venue Manager, you have decided you will meet with a new employee who joined the organization just yesterday and share with them the compliance information that applies to two of the areas in...
-
Tourism is seen by many as the absolute advantage for several countries in the Caribbean. Simpson (2022) states that the Caribbean is the most tourism-dependent region in the world. How can your...

Study smarter with the SolutionInn App


