Specify which of the following equations represent oxidation reduction reactions, and indicate the oxidizing agent, the reducing
Question:
Specify which of the following equations represent oxidation– reduction reactions, and indicate the oxidizing agent, the reducing agent, the species being oxidized, and the species being reduced.
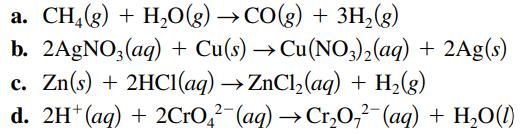
Transcribed Image Text:
a. CH4(g) + H₂O(g) →CO(g) + 3H₂(g) b. 2AgNO3(aq) + Cu(s) → Cu(NO3)₂(aq) + 2Ag(s) c. Zn(s) + 2HCl(aq) → ZnCl₂(aq) + H₂(g) d. 2H+ (aq) + 2CrO²(aq) → Cr₂O₂² (aq) + H₂O(1)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 70% (10 reviews)
a This equation represents a combustion reaction which is a type of oxidationreduction ...View the full answer

Answered By

Labindao Antoque
I graduated in 2018 with a Bachelor of Science degree in Psychology from Dalubhasaan ng Lungsod ng San Pablo. I tutored students in classes and out of classes. I use a variety of strategies to tutor students that include: lecture, discussions about the subject matter, problem solving examples using the principles of the subject matter being discussed in class , homework assignments that are directed towards reinforcing what we learn in class , and detailed practice problems help students to master a concept. I also do thorough research on Internet resources or textbooks so that I know what students need to learn in order to master what is being taught in class .
0.00
0 Reviews
10+ Question Solved
Related Book For 

Chemistry
ISBN: 9781305957404
10th Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Question Posted:
Students also viewed these Engineering questions
-
Which of the following equations represent the correct formula for computing a taxpayer's taxable income Check all that apply. Taxable income = Total income - Exclusions- Adjustments to income...
-
Specify which of the following elements you would expect to have the greatest electron affinity and which would have the least: He, K, Co, S, Cl.
-
Which of the following equations are true for all operators A and B and all functions f and g? (a) (A+ B)f = Af + Bf; (b) A(f + g) = Af + Ag ; (c) (Af)/f = A, provided f 0; (d) AB f = BAf; (e) Af =...
-
You have a net income of $40 000 per year. Your expenses include the following: Rent: $800 per month Insurance: $225 per semi-annually Car Payment: $315 per month Car Expenses: $1 000 per year ...
-
When is the gain on the sale or exchange of securities by a dealer in securities classified as capital gain?
-
Lennox Watches, Inc. makes watches. Its assembly department started the accounting period with a beginning inventory balance of $68,000. During the accounting period, the department incurred $96,000...
-
Consider the inspection described in Example 2.11. From a bin of 50 parts, 6 parts are selected randomly without replacement. The bin contains 3 defective parts and 47 nondefective parts. What is the...
-
International Microcircuits, Inc. Megan Bedding, vice-president of sales for International Microcircuits, Inc. (IM), was delighted when IM was one of the few firms invited to enter a bid to supply a...
-
Physics 521A: Forces Virtual Lab /36 This is a "Virtual Lab". You will be using a free online lab simulation from the University of Colorado Boulder. Go to the following link for the virtual lab:...
-
How is the ctenidium modified from the ancestral form in a typical bivalve?
-
Balance the following oxidationreduction reactions that occur in basic solution. a. Cr(s) + CrO2 (aq) Cr(OH)3(s) b. MnO4 (aq) + S(aq) MnS (s) + S(s) c. CN (aq) + MnO4 (aq) CNO (aq) + MnO (s)
-
Balance the following oxidationreduction reactions that occur in acidic solution using the half-reaction method. a. Cu(s) + NO3(aq) Cu+ (aq) + NO(g) b. CrO72 (aq) + Cl(aq) Cr+ (aq) + Cl(g) c. Pb(s)...
-
The procedure of finding an object by its name is known as (a) search (b) find (c) lookup (d) look for
-
When can an employer legally hire someone not authorized to work in this country?
-
Ron orally engages Dian to act as his agent. During the agency, Ron knows that Dian deals with Mary. Ron also knows that Pete and Brad are aware of the agency but have not dealt with Dian. Ron...
-
Eaters Feast Company is a subsidiary of Food Prep, Inc. A merger of Eaters Feast into Food Prep is a. a corporate liquidation. b. a short-form consolidation. c. a short-form merger. d. a violation of...
-
Grant was the general partner in Oil Build, LP, which he formed to construct offshore oil rigs. He asked Lucinda to invest in the company. A low bridge separated Oil Builds rig construction site from...
-
To obtain a contract with Sadat, Cindy misrepresents that she is a partner with Karl. Karl overhears Cindys misrepresentation but says nothing to Sadat. Cindy breaches the contract. Who is liable to...
-
The unit cell for tin has tetragonal symmetry, with a and b lattice parameters of 0.583 and 0.318 nm, respectively. If its density, atomic weight, and atomic radius are 7.30 g/cm3, 118.69 g/mol, and...
-
When an electric field is applied to a shallow bath of vegetable oil, why do tiny bits of thread floating in the oil align with the field like compasses in a magnetic field?
-
What are the relative values of the chemical shifts observed for nuclei in the spectrometers mentioned in Exercise 15.8a in terms of? (a) Values, (b) Frequencies?
-
The chemical shift of the CH3 protons in diethyl ether is, = 1.16 and that of the CH2 protons is 3.36. What is the difference in local magnetic field between the two regions of the molecule when the...
-
Sketch the appearance of the IH-NMR spectrum of diethyl ether using J = 6.97 Hz and the data in Exercise 15.9b in a spectrometer operating at (a) 350 MHz, (b) 650 MHz.
-
At a minimum, calculate the following Amazon.com ratios for two years the debt to equity ratio current ratio quick ratio and return on equity and net profit margin? Explain what the ratio tells you...
-
For your final assignment, please finish a short reflection of your speaking abilities in various organizational settings that you might find yourself in. Reflect on your strengths and weaknesses as...
-
A maintenance department annually uses 400 units of a specific engineering part and the yearly holding cost per unit is $0.90 along with the setup or ordering cost of $4 per order. Determine the...

Study smarter with the SolutionInn App


