Chlorine gas was first prepared in 1774 by C. W. Scheele by oxidizing sodium chloride with manganese(IV)
Question:
Chlorine gas was first prepared in 1774 by C. W. Scheele by oxidizing sodium chloride with manganese(IV) oxide. The reaction is
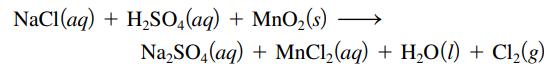
Balance this equation.
Transcribed Image Text:
NaCl(aq) + H₂SO4 (aq) + MnO₂ (s) Na₂SO4 (aq) + MnCl₂(aq) + H₂O(1) + Cl₂(g)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 70% (10 reviews)
To balance this equation you will need to determine the coefficients the numbers in front of the for...View the full answer

Answered By

Vikas Rathour
I have been tutoring for over five years, and I have experience with students of all ages and levels. I have a degree in Education and I am currently working on my Master's degree in Education. I am patient and adaptable, and I am confident that I can help any student improve their grades and confidence in any subject.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Chemistry
ISBN: 9781305957404
10th Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Question Posted:
Students also viewed these Engineering questions
-
Chlorine can be prepared by oxidizing chloride ion (from hydrochloric acid) with potassium dichromate, K2Cr2O7, which is reduced to Cr3+. Write the balanced equation for the reaction.
-
The British chemist Joseph Priestley prepared oxygen in 1774 by heating mercury(II) oxide, HgO. Mercury metal is the other product. If 6.47 g of oxygen is collected, how many grams of mercury metal...
-
The radical reaction of propane with chlorine yields (in addition to more highly halogenated compounds)1-chloropropane and 2-chloropropane. Cl
-
Sita lived in a village and could, not afford refrigerator in her house. She knew how to keep water cold and preserve all perishable items in her house. She kept ivet cloth surrounding the earthen...
-
Amy has LTCGs that are taxed at different tax rates, 15%, 25% and 28%. She also has NSTCLs that amount to less than her NLTCG. The procedure for offsetting the NSTCL against the LTCGs is favorable to...
-
Scientists have experimented with ways of enhancing the oceans ability to absorb atmospheric carbon dioxide. Adding powdered iron to a small plot of the ocean, they found, has the effect of fostering...
-
Give some examples of the variability of sexual behavior. What is considered proper sexual behavior varies greatly among societies.
-
Benedict Company incurred the following costs. 1. Sales tax on factory machinery purchased ........... $ 5,000 2. Painting of and lettering on truck immediately upon purchase ... 700 3. Installation...
-
a) A syrup is a mixture of sugar and water. Syrup A and B are to be mixed together. Syrups A and B contain 3.5 wt% and 4.2 wt% sugar, respectively. The resultant mixture is added with 17.5 kg of pure...
-
Gas absorption or gas scrubbing is a commonly used method for removing environmentally undesirable species from waste gases in chemical manufacturing and combustion processes. The waste gas is...
-
A solution of permanganate is standardized by titration with oxalic acid (H 2 C 2 O 4 ). It required 28.97 mL of the permanganate solution to react completely with 0.1058 g of oxalic acid. The...
-
Balance the following oxidationreduction reactions that occur in basic solution. a. Cr(s) + CrO2 (aq) Cr(OH)3(s) b. MnO4 (aq) + S(aq) MnS (s) + S(s) c. CN (aq) + MnO4 (aq) CNO (aq) + MnO (s)
-
One design challenge for a capacitor-powered electric car is that the voltage would change with time as the capacitors discharged. If the capacitors in a car were discharged to half their initial...
-
Shore Co. records its transactions in U.S. dollars. A sale of goods resulted in a receivable denominated in Japanese yen, and a purchase of goods resulted in a payable denominated in euros. Shore...
-
Joel is a shareholder in an S corporation. He purchased 200 shares of the corporation at the beginning of the year for $25,000. His Schedule K-1 (Form 1120-S) for the same year reported ordinary...
-
Susan paints portraits of animals that she hangs in her home, an activity she characterizes as a hobby. Recently, a friend noticed the pictures and asked Susan to paint a portrait of her dog for...
-
If you deposit $400 each year for 8 years in an account that pays 7%, How much will you have at the end of the 8 years? Use the TVM keys on your calculator... NOT the tables in the book Carry answer...
-
8 . For the season, the accounting department forwarded to you the following information. Construct a P&L Statement from the following figures. Calculate the TOTAL COST OF GOODS SOLD considering...
-
Kimberly Manis, an architect, organized Manis Architects on January 1, 2016. During the month, Manis Architects completed the following transactions: a. Issued common stock to Kimberly Manis in...
-
State whether each of the following will increase or decrease the power of a one-way between-subjects ANOVA. (a) The effect size increases. (b) Mean square error decreases. (c) Mean square between...
-
In our discussion of blackbody radiation, the average energy of an oscillator E osc = hv/(e hv/kBT 1) was approximated as E osc = hv/[(1 + hv/k B T) 1] = k B for hv/k B T < < 1. Calculate the...
-
Using the root mean square speed, v rms = (v 2 ) 1/2 = 3k B T/m, calculate the gas temperatures of He and Ar for which = 0.25 nm, a typical value needed to resolve diffraction from the surface of a...
-
Electrons have been used to determine molecular structure by diffraction. Calculate the speed and kinetic energy of an electron for which the wavelength is equal to a typical bond length, namely,...
-
Watch the video "IDEO shopping cart project https://www.youtube.com/watch?time_continue=5&v=M66ZU2PCIcM what are the attributes in the IDEO organization that encourage and enhance innovation in an...
-
1 Critically evaluate the importance of the media in influencing partners, opinion formers, stakeholders, customers and the public at large. 2 Critically assess domestic, national and international...
-
A 0.7 MeV photon scatters from an electron initially at rest. If the photon scatters at an angle of 35, calculate (a) the energy and wavelength of the scattered photon, (b) the kinetic energy of the...

Study smarter with the SolutionInn App


