Balance the following oxidationreduction reactions that occur in basic solution. a. Cr(s) + CrO2 (aq) Cr(OH)3(s)
Question:
Balance the following oxidation–reduction reactions that occur in basic solution.
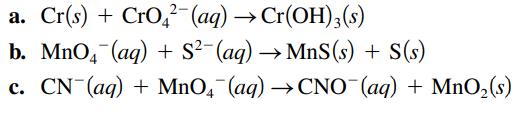
Transcribed Image Text:
a. Cr(s) + CrO2 (aq) → Cr(OH)3(s) b. MnO4 (aq) + S²(aq) →MnS (s) + S(s) c. CN (aq) + MnO4 (aq) →CNO (aq) + MnO₂ (s)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (10 reviews)
a Crs CrO4aq 8OHaq CrOH3s 4H2Ol b MnO4 aq S2 aq 2OHaq MnS s Ss H2Ol c CN aq MnO4 aq ...View the full answer

Answered By

Labindao Antoque
I graduated in 2018 with a Bachelor of Science degree in Psychology from Dalubhasaan ng Lungsod ng San Pablo. I tutored students in classes and out of classes. I use a variety of strategies to tutor students that include: lecture, discussions about the subject matter, problem solving examples using the principles of the subject matter being discussed in class , homework assignments that are directed towards reinforcing what we learn in class , and detailed practice problems help students to master a concept. I also do thorough research on Internet resources or textbooks so that I know what students need to learn in order to master what is being taught in class .
0.00
0 Reviews
10+ Question Solved
Related Book For 

Chemistry
ISBN: 9781305957404
10th Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Question Posted:
Students also viewed these Engineering questions
-
Balance each of the following oxidation reduction reactions by using the oxidation states method. a. C2H6(g) + O2(g) CO2(g) + H2O(g) b. Mg(s) + HCl(aq) Mg2+(aq) + Cl2(aq) + H2(g) c. Cu(s) + Ag+(aq)...
-
Balance these redox reactions that occur in aqueous solution. Use whatever water-derived species is necessary; there may be more than one correct balanced equation. a. CrO3 + Ni2+ ( Cr3+ + Ni3+ b....
-
Balance these redox reactions that occur in aqueous solution. Use whatever water-derived species is necessary; there may be more than one correct balanced equation. a. ClO + Ti2+ ( Ti4+ + Cl b. BrO3...
-
You have the following information on two bonds Bond 1 Bond 1 Type Coupon Zero Coupon Term (Yrs) 3 3 Payment Frequency Annual Annual Face Amount ($) $25,000 $25,000 Coupon (%) 6% NA a. Calculate the...
-
In 2002, Florence purchased 30 acres of land. She has not used the land for business purposes or made any substantial improvements to the property. During the current year, she subdivides the land...
-
Rank the following organic molecules in order of increasing solubility in water: (b) (c) (a)
-
How does cross-cultural evidence raise questions about the division of humanity into male and female?
-
Consuelo Chua, Inc., is a disk drive manufacturer in need of an aggregate plan for July through December. The company has gathered the following data: COSTS Holding cost.................$8/disk/month...
-
Varsity Furniture began June with merchandise inventory of 41 sofas that cost a total of $29,110. During the month, the company purchased and sold merchandise on account as follows: June 7 June 14...
-
The client received a large shipment of inventory on December 31st during the inventory observation. What procedures should the auditor perform to verify that the acquisition of inventory is properly...
-
Chlorine gas was first prepared in 1774 by C. W. Scheele by oxidizing sodium chloride with manganese(IV) oxide. The reaction is Balance this equation. NaCl(aq) + HSO4 (aq) + MnO (s) NaSO4 (aq) +...
-
Specify which of the following equations represent oxidation reduction reactions, and indicate the oxidizing agent, the reducing agent, the species being oxidized, and the species being reduced. a....
-
How many outs are there in a baseball game that lasts the full 9 innings?
-
Cultural competency is an evolving process of interrelated cultural and healthcare factors. We explored various issues of health disparities and characteristics that affect the ability to develop a...
-
The balance sheet of Mega Company includes the following accounts: Accounts Receivable $25,000, Goodwill $7,000, Furniture $5,000, Cash $8,000, Supplies $11,000, Equipment $14,000, patents $3,000 and...
-
Suggest and justify an appropriate launch strategy for Mh's. What organizational capabilities will be critical to Mh's success?
-
Data analytics and predictive modeling have revolutionized the field of managerial auditing, providing auditors with powerful tools to extract insights, detect patterns, and make informed decisions....
-
A firm has three categories of budgeted overhead costs. Supervisor Salaries: $ 7 5 0 , 0 0 0 Equipment Lease: $ 1 9 0 , 0 0 0 Maintenance: $ 7 5 , 0 0 0 After an activity - based costing study, the...
-
On January 7, 2016, Captec Company purchased $4,175 of supplies on account. In Captec Company's chart of accounts, the supplies account is No. 15, and the accounts payable account is No. 21. a....
-
A researcher reports a significant two-way between-subjects ANOVA, F(3, 40) = 2.96. State the decision to retain or reject the null hypothesis for this test.
-
Calculate the magnetic field needed to satisfy the resonance condition for unshielded protons in a 150.0 MHz radiofrequency field.
-
Use Table 15.2 to predict the magnetic fields at which (a) 14N, (b) 19F, and (c) 31p comes into resonance at (i) 300 MHz, (ii) 750 MHz.
-
Calculate the relative population differences (oN/N) for 13Cnuclei in fields of (a) 0.50 T, (b) 2.5 T, and (c) 15.5 T at 25C.
-
Doing a strategic analysis of GraceKennedy Limited, What is the current level of its economic performance, an indication of the factors responsible for the current performance and recommendations for...
-
How would you evaluate Cisco social media marketing success? Outline which, if any, of the mentioned media plans Cisco should pursue. Why? What are the challenges of using social media? Do you have...
-
Office Ltd manufactures furniture for computer work stations. The company uses a job costing system. Manufacturing overhead is applied to production at a predetermined overhead rate of $60 per direct...

Study smarter with the SolutionInn App


