Consider a galvanic cell based on the following theoretical half-reactions: What is the value of G
Question:
Consider a galvanic cell based on the following theoretical half-reactions:
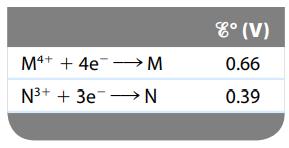
What is the value of ΔGο and K for this cell?
Transcribed Image Text:
M4+ + 4e→→→→→M N³+ + 3e N 8° (V) 0.66 0.39
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 62% (8 reviews)
Its under conditions as well as the equilibrium constant K We know that this is a galvanic cell and ...View the full answer

Answered By

Dudhat Vaidehi
I tutored mostly elementary school students privately after school and during the summer. We met in their homes or at the public library. I charged an hourly fee, and I provided any necessary materials.
Having taught in special education in two local schools for many years meant that I had contact with a lot of parents of special needs students. I never had to advertise — word of mouth was how most folks knew of me. At one point I did have a website, but didn't utilize it much. I stayed very busy, especially in the summers, and always had a full schedule. I typically met with each student's teacher in order to get an idea of what the focus of my instruction/remediation should be. Becoming familiar with the student's learning style(s) was also very helpful. Often parents would share records and test results with me. After each tutoring session, I documented the student’s progress and gave parents written updates, as well as phone calls or emails as needed.
While my students and I certainly utilized technology and the internet often during our sessions, I never tutored online or for any tutoring company, so am not familiar with the curriculums or methods used in those settings.
Tutoring one on one was very enjoyable and rewarding. My students and I had fun, and grew quite fond of one another. The extra income was a bonus. I had to retire from tutoring due to a physically handicapping disease, and miss my students very much.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Chemistry
ISBN: 9781305957404
10th Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Question Posted:
Students also viewed these Engineering questions
-
Consider the standard galvanic cell based on the following half reactions Cu2+ + 2e- Cu Ag+ + e- Ag The electrodes in this cell are Ag(s) and Cu(s). Does the cell potential increase, decrease, or...
-
Consider the galvanic cell based on the following halfreactions: b. Calculate ÎGo and K for the cell reaction at 25oC. c. Calculate cell at 25oC when [Zn2+] = 0.10 M and [Fe2+] = 1.0 Ã...
-
Consider the galvanic cell based on the following halfreactions: b. Calculate ÎGo and K for the cell reaction at 25oC. c. Calculate cell at 25oC when [Au3+] = 1.0 Ã 102 M and [Tl+] = 1.0...
-
Route Canal Shipping Company has the following schedule for aging of accounts receivable: AGE OF RECEIVABLES APRIL 30, 2001 a. Fill in column (4) for each month. b. If the firm had $1,440,000 in...
-
Link from the text Web site to the online Economic Report of the President and get annual data on real GDP and real investment. Calculate the growth rates of the two variables for each year since...
-
A 10.0-kg box is palled by a horizontal wire in a circle on a rough horizontal surface for which the coefficient of kinetic friction is 0.250. Calculate the work done by friction during one complete...
-
Determine the convection heat transfer coefficient, thermal resistance for convection, and the convection heat transfer rate that are associated with air at atmospheric pressure in cross flow over a...
-
A production line operation is tested for filling weight accuracy using the following hypotheses. Hypothesis Conclusion and Action H0: = 16 .....Filling okay; keep running Ha: 16 ..... Filling off...
-
Without invoking Java, given the following Java program fragment Scanner console new Scanner (System.in); int int 12 count: int evenSum int oddSum; 107 evenSum odds um 0; 0: for (count...
-
1. After examining financial data for a monthly period, the owner of a small business expressed surprise that the firms cash balance had decreased during the month even though there was substantial...
-
When the environment is contaminated by a toxic or potentially toxic substance (for example, from a chemical spill or the use of insecticides), the substance tends to disperse. How is this consistent...
-
In the electrolysis of a sodium chloride solution, what volume of H 2 (g) is produced in the same time it takes to produce 257 L Cl 2 (g), with both volumes measured at 50. C and 2.50 atm?
-
Show that the work done by a constant force field F = ai + bj + ck in moving a particle along any path from A to B is W = F.AB.
-
A company's common stock pays a dividend of $ 5 0 each year. The next dividend will be paid in one year. It will be $ 5 0 . If the dividend is expected to increase at 5 % per year thereafter,...
-
George is upset by a recent Ontario law that prohibits him from owning a "dangerous dog". George hired a well-known lawyer to challenge the law under the Canadian Charter of Rights and Freedom....
-
How do transformational leaders leverage emotional intelligence and empathetic communication to inspire followership and foster collective commitment to strategic objectives?
-
[3] In case of uniformly moving charge, show that the ratio of the electric field perpendicular in the direction of charge motion to that parallel in the direction of charge motion is 1/y.
-
Below is an example of an uneven cash flow problem. In an uneven cash flow problem the cash flows occur over time, but they are not equal and they are not necessarily periodic like an annuity. These...
-
A communications link in a network should be provided with a backup link if its failure makes it impossible for some message to be sent. For each of the communications networks shown here in (a) and...
-
Audrey purchases a riding lawnmower using a 2-year, no-interest deferred payment plan at Lawn Depot for x dollars. There was a down payment of d dollars and a monthly payment of m dollars. Express...
-
Compound F, a hydrocarbon with M + = 96 in its mass spectrum, undergoes reaction with HBr to yield compound G. Propose structures for F and G, whose 13 C NMR spectral data follow. Compound 1:...
-
3-Methyl-2-butanol has five signals in its 13C NMR spectrum at 17.90, 18.15, 20.00, 35.05, and 72.75 ?. Why are the two methyl groups attached to C3 nonequivalent? Making a molecular model should be...
-
A 13C NMR spectrum of commercially available 2, 4-pentanediol, shows five peaks at 23.3, 23.9, 46.5, 64.8, and 68.1 ?. Explain. CHCH-CHCH3 2,4-Pentanediol
-
What is one specific security tactics to protect the network. Identify specific tools selections to accomplish compliance and audit requirement Describe how alerts would be collected and presented...
-
1. Solve each equation for x, where 0x & a) sin x-= 0 b) cosx-3 cosx+2=0
-
Explain Article III Standing, and explain how to achieve standing. b) Explain Negligence, and provide an example of a Negligence claim. c) What happened in the case of Genesco v. Visa? d) Explain the...

Study smarter with the SolutionInn App


