Consider Figure 11-5. For an equal component alloy of A and B starting at 1400C, write each
Question:
Consider Figure 11-5. For an equal component alloy of A and B starting at 1400°C, write each reaction as the crucible is cooled to room temperature (25°C).
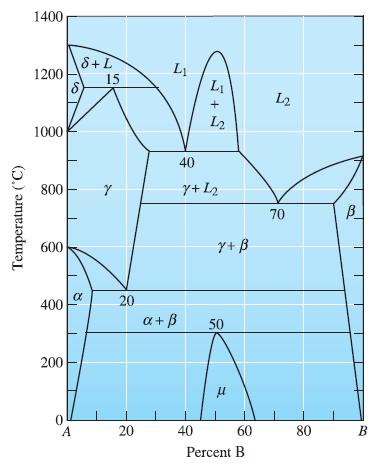
Transcribed Image Text:
Temperature (°C) 1400 1200 1000 800 600 400 200 0 S A S+L α 15 Y 20 20 Li a + ß L₁ + 12 40 7+12 40 Y+ B 50 μ 60 Percent B L2 70 80 B
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (8 reviews)
Reactions are as follows 1 L 1 930 0 C ...View the full answer

Answered By

User l_831638
Hello! My name is Sadhana.M.S. I made specilization in Nuclear Physics at Kuvempu university.I've been an physics tutor for degree level student . My motto is "teach student how to learn".
0.00
0 Reviews
10+ Question Solved
Related Book For 

The Science And Engineering Of Materials
ISBN: 9781305076761
7th Edition
Authors: Donald R. Askeland, Wendelin J. Wright
Question Posted:
Students also viewed these Engineering questions
-
For what types of A and B is the simple assignment statement A = B legal in C++ but not Java?
-
For the separation of A and B by column 22 in the previous problem: a. If broadening is mainly due to longitudinal diffusion, how should the flow rate be changed to improve resolution? b. If...
-
Before any reaction occurs, the concentrations of A and B in the reaction below are each 0.03900 M . What is the equilibrium constant if the concentration of A at equilibrium is 0.01365 M ? 2A(aq) +...
-
Can public works increase equilibrium wages?
-
Calculate the forward discount or premium for the following spot and three-month forward rates: (a) SR = SF2/1 and SF2.02/1 where SF is the Swiss franc and is the euro (b) SR = 200/$1 and FR = 190/$1
-
Hana Coffee Company roasts and packs coffee beans. The process begins by placing coffee beans into the Roasting Department. From the Roasting Department, coffee beans are then transferred to the...
-
Discuss the historical role of HR benchmarking and its strengths and weaknesses as part of a metrics and analytics program in organizations today.
-
Assume you are the manufacturer of modular office systems and furniture as well as office organization elements (desktop and wall organizers, filing systems, etc.). Your company has been asked to...
-
Lethe has borrowed $100 000 to purchase a drilling machine and promise to make 5 years of annual repayments at a fixed interest rate of 11.5% p.a. What is the annual repayment?
-
Nearly There (the Company), an SEC registrant, designs, develops, manufactures, and sells various navigation products and services. Because of significant research and development expenses and...
-
Consider a Pb-70% Sn alloy. Determine (a) If the alloy is hypoeutectic or hypereutectic; (b) The composition of the first solid to form during solidification; (c) The amounts and compositions of each...
-
Determine the phases that are present and the compositions for each phase in Cu-55 wt% Ag at 600C. Data Form Problem 11-23 Consider a Pb-70% Sn alloy. Determine (a) If the alloy is hypoeutectic or...
-
How do peoples images or perceptions of tourism destinations affect their decisions on where to travel?
-
New Zealand rewrote the charter of its central bank in the early 1990s to make low inflation its only goal. Why would New Zealand want to do this?
-
Suppose the government amends the constitution to prevent government officials from negotiating with terrorists. What are the advantages of such a policy? What are the disadvantages?
-
The legislation governing the Federal Reserve Board The 1977 Federal Reserve Act, as amended in 1978, 1988, and 2000 governs the behavior of the Federal Reserve. a. In your opinion, does this excerpt...
-
Chinese economic growth is the outstanding feature of the world economic scene over the past two decades. a. In 2014, U.S. output was $17.4 trillion, and Chinese output was $10.4 trillion. Suppose...
-
Suppose you have been elected to Congress. One day, one of your colleagues makes the following statement: The Fed chair is the most powerful economic policy maker in the United States. We should not...
-
Find Leq in the circuit in Fig. 6.78. Lea ell ele
-
Define cultural intelligence. Cite the books or journal articles you found in Capella's library. Explain why cultural intelligence is important for HR practitioners and other organizational managers.
-
Is it possible to precipitate 99.0% of 0.010 M Ce 3+ by adding oxalate (C 2 O 4 2- ) without precipitating 0.010 M Ca 2+ ? CaC204 Ksp = 1.3 x 10-8 %3D Ce,(C,O4)3 Ksp 5.9 X 10-30
-
For a solution of Ni 2+ and ethylenediamine, the following equilibrium constants apply at 20C: Calculate the concentration of free Ni 2+ in a solution prepared by mixing 0.100 mol of en plus 1.00 mL...
-
If each compound is dissolved in water, will the solution be acidic, basic, or neutral? (a) Na Br (e) (CH3),N CI (b) Na CH,CO, (f) (CH,),N*O-co, (c) NH CI (g) Fe(NO3)3 (d) K3PO4
-
What is the length of the undertaking of a person who sponsors their common-law partner?
-
2. (a) (5pts) A 3-year zero-coupon bond has face value (PAR) = $1,000 and price = $729. What is the yield to maturity of this bond? (b) (5pts) Suppose that when the issuer of the bond goes bankrupt,...
-
Suppose that over one year the number of Dollars per one Euro changes from 1.00 to 1.20. That is, the dollar depreciates. Obtain the Annual depreciation rate of the Dollar. Obtain the Annualized...

Study smarter with the SolutionInn App


