Consider the following idealized PES spectrum for potassium: Explain the location and relative intensities of the various
Question:
Consider the following idealized PES spectrum for potassium:
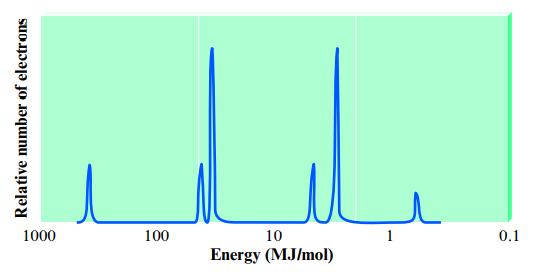
Explain the location and relative intensities of the various peaks.
Transcribed Image Text:
Relative number of electrons 1000 100 10 Energy (MJ/mol) 0.1
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (5 reviews)
Potassium has an atomic number of 19 The electronic setup of it is Orbitals co...View the full answer

Answered By

Aketch Cindy Sunday
I am a certified tutor with over two years of experience tutoring . I have a passion for helping students learn and grow, and I firmly believe that every student has the potential to be successful. I have a wide range of experience working with students of all ages and abilities, and I am confident that I can help students succeed in school.
I have experience working with students who have a wide range of abilities. I have also worked with gifted and talented students, and I am familiar with a variety of enrichment and acceleration strategies.
I am a patient and supportive tutor who is dedicated to helping my students reach their full potential. Thank you for your time and consideration.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Chemistry
ISBN: 9781305957404
10th Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Question Posted:
Students also viewed these Engineering questions
-
Consider the following idealized PES spectrum for carbon: Explain the location and relative intensities of the various peaks. Relative number of electrons 100 10 Energy (MJ/mol) 1 0.1
-
Consider the following idealized PES spectrum for an element: What is the identity of the element? Relative number of electrons 1000 100 10 Energy (MJ/mol) 0.1
-
Consider the following idealized PES spectrum for an element: What is the identity of the element? Explain the relative positions of the various peaks. Relative number of electrons 1000 100 t 1 10...
-
Pew Research published survey results from two random samples. Both samples were asked, Have you listened to an audio book in the last year? The results are shown in the table below. a. Find and...
-
"Part 1: The Performance Lawn Equipment database contains data needed to develop a pro forma income statement. Dealers selling PLE products all receive 18% of sales revenue for their part of doing...
-
Monroe Marketing is considering employing another marketing consultant to help with managing its clients. The human resources manager of the firm is weighing up whether to employ a marketing graduate...
-
Consider the incompressible, two-dimensional flow of a nonviscous fluid between the boundaries shown in Fig. P6.22. The velocity potential for this flow field is \[ \phi=x^{2}-y^{2} \] (a) Determine...
-
Stylz Company, a recent start-up fashion retailer based in the United States, is deciding between opening its first sales presence in either Italy's Tuscany Region or Spain's Matarrana Region....
-
Given: 1kg = 1000g 1. A pitcher threw a pitch that generated an impulse of 3.25 Ns on the catcher's glove. If the mass of the baseball is 142g, what was the velocity of the pitched baseball when it...
-
An inclined manometer is a useful device for measuring small pressure differences. The formula given in Section 3.4 for the pressure difference in terms of the liquid-level difference h remains...
-
Give a possible set of values of the four quantum numbers for all the electrons in a boron atom and a nitrogen atom if each is in the ground state.
-
How many 4d electrons would be predicted in the ground state for the following elements? a. Zirconium b. Cadmium c. Iridium d. Iron
-
The transfer function of the network is given by the expression Determine the damping ratio, the un-damped natural frequency, and the type of response that will be exhibited by the network. G(s) =...
-
Paradiddle Dance Paradise is saving up for an expansion. If they set aside $ 4 , 0 0 0 at the beginning of each quarter for the next 6 years in an account that pays 4 . 4 % compounded quarterly, how...
-
Frogs have been breeding like flies at the Enormous State University (ESU) campus! Each year, the pledge class of the Epsilon Delta fraternity is instructed to tag all the frogs residing on the ESU...
-
What is the future value of $ 9 0 , 3 8 0 . 0 0 given an interest rate of 1 7 . 0 0 % compounded quarterly for 3 7 years?
-
Analysts expect Oscorp Industries to make payouts of $2.28B at the end of this year. Assume that all payouts occur annually at the end of the year and that we are at the beginning of the year....
-
Write a program that asks the user to type in a time in seconds and display it in the standard format hours: minutes: seconds (where 1 hour 3600 seconds and 1 minute = 60 seconds). Your output should...
-
The solubility of CO2 in water at 25C and 1 atm is 0.034 mol/L. What is its solubility under atmospheric conditions? (The partial pressure of CO2 in air is 0.0003 atm.) Assume that CO2 obeys Henry's...
-
The polar coordinates of a point are given. Find the rectangular coordinates of the point. (-1, - /3)
-
Draw all the cis-trans isomers for these compounds: (a) CH 3 CH = CHCH = CHCH 2 CH 3 (b) CH 3 CH = CHCH = CHCH 3 (c) CH 3 CH = CHCH = CH 2
-
Which of these groups has the higher priority? Br b) -C=N or -CH,CH2 a) -C=CH or CH3 CH3 c) -C=CH2 or -C-CH3 CH3 -C-CH3 d) -C=CH or CH3
-
Assign the configurations of these compounds as Z or E: 3 CH,CH2CH3 C=C CH,Br C=C HOCH,CH2 b) a) H,NCH, CH,CHCH3 CH,CH CH3 H3 - HO- Ph c) d)
-
Speculate sales : Speculate on the projected sales. Justify your product or service by the numbers; discuss your projected revenue gain. Note: it MUST have an ROI that justifies the project for...
-
8.6 In Figure P8.6 the experimental unit step response of a process system is shown. (a) What is the transfer function that can be used to best describe the system dynamics among the following. 1 (1)...
-
An 84 year old female arrives to the ER today with her son with a complaint of new onset of confusion. What are your differential diagnoses for a patient with new altered mental status? Go through...

Study smarter with the SolutionInn App


