Determine the carburizing time necessary to achieve a carbon concentration of 0.45 wt% at a position 2-mm
Question:
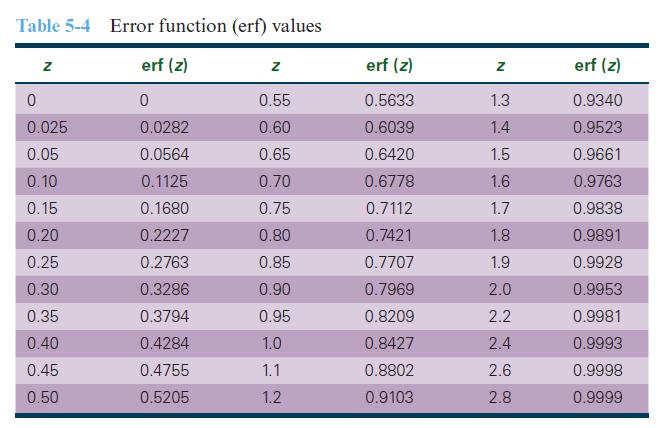
Determine the carburizing time necessary to achieve a carbon concentration of 0.45 wt% at a position 2-mm into a steel alloy that initially contains 0.20 wt% carbon. The surface concentration is to be maintained at 1.30 wt% at 1000°C. Use Table 5-4 for error function values as needed.
Transcribed Image Text:
Table 5-4 Error function (erf) values erf (z) 0 0.0282 0.0564 0.1125 0.1680 0.2227 0.2763 0.3286 0.3794 0.4284 0.4755 0.5205 Z 0 0.025 0.05 0.10 0.15 0.20 0.25 0.30 0.35 0.40 0.45 0.50 Z 0.55 0.60 0.65 0.70 0.75 0.80 0.85 0.90 0.95 1.0 1.1 1.2 erf (z) 0.5633 0.6039 0.6420 0.6778 0.7112 0.7421 0.7707 0.7969 0.8209 0.8427 0.8802 0.9103 Z 1.3 1.4 1.5 1.6 1.7 1.8 1.9 2.0 2.2 2.4 2.6 2.8 erf (z) 0.9340 0.9523 0.9661 0.9763 0.9838 0.9891 0.9928 0.9953 0.9981 0.9993 0.9998 0.9999
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 93% (15 reviews)
The carburizing time necessary is called formula3 and is g...View the full answer

Answered By

Brian Otieno
I'm Brian , an experienced professional freelancer with countless hours of success in freelancing many subjects in different disciplines. Specifically, I have handled many subjects and excelled in many disciplines. I have worked on many Computer Science projects and have been able to achieve a lot in that field. Additionally, I have handled other disciplines like History, Humanities, Social Sciences, Political science, Health care and life science, and Religion / Theology. My experience generally in these subjects has made me able to deliver high-quality projects in a very timely fashion. I am very reliable at my job and will get the work done in time, no matter what. In Addition, I have managed to ensure that the work meets my client's expectations and does not cause an error. I am a hard-working and diligent person who is highly responsible for everything I do. Generally, Freelancing has made me more accountable for doing my job. Additionally, I have had a passion for writing for the last seven years in this field.
0.00
0 Reviews
10+ Question Solved
Related Book For 

The Science And Engineering Of Materials
ISBN: 9781305076761
7th Edition
Authors: Donald R. Askeland, Wendelin J. Wright
Question Posted:
Students also viewed these Engineering questions
-
Determine the carburizing time necessary to achieve a carbon concentration of 0.45 wt% at a position 2 mm into an iron-carbon alloy that initially contains 0.20 wt% C. The surface concentration is to...
-
Determine the carburizing time necessary to achieve a carbon concentration of 0.30 wt% at a position 4-mm into a steel alloy that initially contains 0.10 wt% carbon. The surface concentration is to...
-
Determine the carburizing time necessary to achieve a carbon concentration of 0.30 wt% at a position 4 mm into an iron-carbon alloy that initially contains 0.10 wt% C. The surface concentration is to...
-
A. Discuss the following conceptual model/framework of Orem's Self-Care Theory CONDITIONING FACTORS (12 MARKS) Orem's Self-Care Theory Conceptual Framework SELF-CARE AGENCY SELF-CARE DEFICIT NURSING...
-
What does limited liability mean?
-
A CI is desired for the true average stray-load loss (watts) for a certain type of induction motor when the line current is held at 10 amps for a speed of 1500 rpm. Assume that stray-load loss is...
-
Help for Insomniacs In Exercise 1.23, we learned of a study to determine whether just one session of cognitive behavioral therapy can help people with insomnia. In the study, forty people who had...
-
Goldencoast Industries uses standard costing and a flexible budget for cost planning and control. Its monthly budget for overhead costs is $100,000 of fixed costs plus $5 per machine hour. Monthly...
-
Fairfield Company's raw materials inventory transactions for the most recent month are summarized here: Note: Assume, purchase of raw materials is on account. Beginning raw materials Purchases of raw...
-
According to a study done by Nick Wilson of Otago University Wellington, the probability a randomly selected individual will not cover his or her mouth when sneezing is 0.267. Suppose you sit on a...
-
Nitrogen from a gaseous phase is to be diffused into pure iron (BCC) at 700C. If the surface concentration is maintained at 0.10 wt% N, what will the concentration 1 mm from the surface be after 10...
-
Certain ceramics with special dielectric properties are used in wireless communication systems. Barium magnesium tantalate (BMT) and barium zinc tantalate (BZT) are examples of such materials....
-
A study reported by Griffin et al. compared the rate of pneumonia in 19971999 before pneumonia vaccine (PCV7) was introduced and in 20072009 after pneumonia vaccine was introduced. Read the excerpts...
-
Marguerite Eaton and Bobby Joe Waldrop moved into a mobile home on land owned by her son, James. Bobby Joe asked James to transfer that portion of the land to him and Marguerite, stating falsely that...
-
Lisa Goldstein reserved space for a marriage ceremony in a building owned by Orensanz Events, LLC, in New York City. The rental agreement provided that on cancellation of the event for any reason...
-
Vals Foods signs a contract to buy 1,500 pounds of basil from Sun Farms, a small organic herb grower, if an independent organization inspects the crop and certifies that it contains no pesticide or...
-
Evelyn Vollmer orally agreed to loan Danny Lang $150,000 to make an investment in a local nightclub. The loan was to be repaid from the profits received from the investment. Their agreement was never...
-
Chelene had been a caregiver for Martas elderly mother, Janis, for nine years. Shortly before Janis passed away, Chelene convinced her to buy Chelenes house for Marta. Janis died before the papers...
-
Using PSpice, find the transmission parameters for the network in Fig. 19.126. 2 2
-
B.) What is the approximate concentration of free Zn 2+ ion at equilibrium when 1.0010 -2 mol zinc nitrate is added to 1.00 L of a solution that is 1.080 M in OH - . For [Zn(OH) 4 ] 2- , K f = 4.610...
-
Consider the following space-filling models for dry ice, ethanol, and caffeine: What amount (moles) is represented by each of the following samples? a. 1.50 g of dry ice b. 2.72 10 21 molecules of...
-
Prevacid is used to treat gastroesophageal reflux disease (GERD). The chemical formula of Prevacid is C 16 H 14 F 3 N 3 O 2 S. a. What is the molar mass of Prevacid? b. What mass of fluorine is in...
-
Freon-12 (CCl 2 F 2 ) is used as a refrigerant in air conditioners and as a propellant in aerosol cans. Calculate the number of molecules of Freon-12 in 5.56 mg of Freon-12. What is the mass of...
-
During the 2017 baseball season in a game against the Texas Rangers, Matt Bush (B) and Joey Gallo (A) collided while trying to catch an infield fly. Assume that they both started from rest at the...
-
= cx(b - x), Given: A particle P travels on a path described by the Cartesian coordinates of y where x and y have the units of meters. The x-component of velocity, x, for P is constant. Find: For...
-
Q3 (15p) A schematic of thermal boundary layer development on an isothermal flat plate with a constant free stream flow velocity (u) is shown below. The plate is made of pure iron and the flow is...

Study smarter with the SolutionInn App


