Nitrogen from a gaseous phase is to be diffused into pure iron (BCC) at 700C. If the
Question:
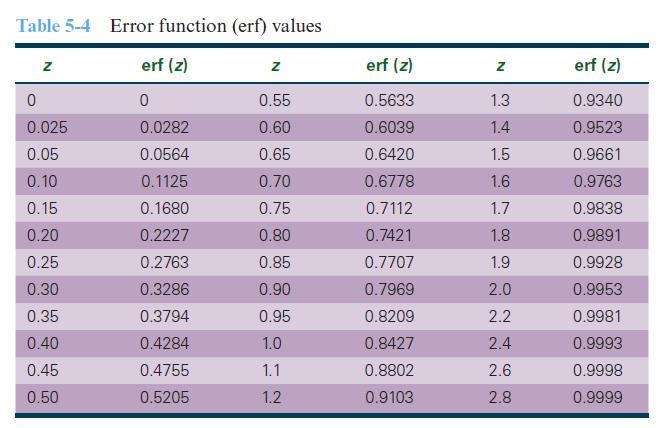
Nitrogen from a gaseous phase is to be diffused into pure iron (BCC) at 700°C. If the surface concentration is maintained at 0.10 wt% N, what will the concentration 1 mm from the surface be after 10 hrs? Use Table 5-4 for error function values as needed.
Transcribed Image Text:
Table 5-4 Error function (erf) values erf (z) 0 0.0282 0.0564 0.1125 0.1680 0.2227 0.2763 0.3286 0.3794 0.4284 0.4755 0.5205 Z 0 0.025 0.05 0.10 0.15 0.20 0.25 0.30 0.35 0.40 0.45 0.50 Z 0.55 0.60 0.65 0.70 0.75 0.80 0.85 0.90 0.95 1.0 1.1 1.2 erf (z) 0.5633 0.6039 0.6420 0.6778 0.7112 0.7421 0.7707 0.7969 0.8209 0.8427 0.8802 0.9103 Z 1.3 1.4 1.5 1.6 1.7 1.8 1.9 2.0 2.2 2.4 2.6 2.8 erf (z) 0.9340 0.9523 0.9661 0.9763 0.9838 0.9891 0.9928 0.9953 0.9981 0.9993 0.9998 0.9999
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 36% (11 reviews)
The concentration 1 mm from the surface be after 10 hrs 01010 10...View the full answer

Answered By

Brian Otieno
I'm Brian , an experienced professional freelancer with countless hours of success in freelancing many subjects in different disciplines. Specifically, I have handled many subjects and excelled in many disciplines. I have worked on many Computer Science projects and have been able to achieve a lot in that field. Additionally, I have handled other disciplines like History, Humanities, Social Sciences, Political science, Health care and life science, and Religion / Theology. My experience generally in these subjects has made me able to deliver high-quality projects in a very timely fashion. I am very reliable at my job and will get the work done in time, no matter what. In Addition, I have managed to ensure that the work meets my client's expectations and does not cause an error. I am a hard-working and diligent person who is highly responsible for everything I do. Generally, Freelancing has made me more accountable for doing my job. Additionally, I have had a passion for writing for the last seven years in this field.
0.00
0 Reviews
10+ Question Solved
Related Book For 

The Science And Engineering Of Materials
ISBN: 9781305076761
7th Edition
Authors: Donald R. Askeland, Wendelin J. Wright
Question Posted:
Students also viewed these Engineering questions
-
Nitrogen from a gaseous phase is to be diffused into pure iron at 700C. If the surface concentration is maintained at 0.1 wt% N, what will be the concentration 1 mm from the surface after 10 h? The...
-
Nitrogen from a gaseous phase is to be diffused into pure iron at 700C. If the surface concentration is maintained at 0.1 wt% N, what will be the concentration 1 mm from the surface after 10 h? The...
-
Nitrogen from a gaseous phase is to be diffused into pure iron at 675C. If the surface concentration is maintained at 0.2 wt% N, what will be the concentration 2 mm from the surface after 25 h? The...
-
Selected information from Indigo Books & Music Inc.'s income statements for three recent years follows (dollars in thousands): Instructions (a) Calculate gross profit, and profit from operations, for...
-
What is a NUANS report, and what is its purpose?
-
A normal probability plot of the n = 26 observations on escape time given in Exercise 36 of Chapter 1 shows a substantial linear pattern; the sample mean and sample standard deviation are 370.69 and...
-
Success at Quitting Smoking After six months, 156 of the 914 smokers who accepted the invitation to be in the reward-only program stopped smoking, while 78 of the 146 smokers who paid a deposit quit....
-
Consider Wal-Mart, a large retailer. Classify the following items as an Asset (A), a Liability (L), or Stockholders Equity (S) for Wal-Mart: a . _____ Accounts payable b. _____ Common stock c. _____...
-
Campbell, a single taxpayer, earns $400,000 in taxable income and $2,000 in interest from an investment in State of New York bonds. (Use the U.S. tax rate schedule.) Required: a. How much federal tax...
-
Details 1. Skill Builder Exercise - Working with PRTK This exercise requires the Mantooth and Washer case. Open the Mantooth case and Washer Case. Identify the encrypted files. Export the following...
-
One method of fabricating complex aerospace components is diffusion bonding. We wish to join a nickel sheet, intended to rapidly transfer lightning strike data away from critical locations, to an FCC...
-
Determine the carburizing time necessary to achieve a carbon concentration of 0.45 wt% at a position 2-mm into a steel alloy that initially contains 0.20 wt% carbon. The surface concentration is to...
-
On January 1, Year 2, several supporters of the organization spent their own money to construct a garage for its vehicles that is worth $70,000. It should last for 10 years and will have no salvage...
-
Internet giant Zoidle, a U.S. company, generated sales of 2.5 billion in the United Kingdom (UK) in 2013 (roughly $4 billion in U.S. dollars). The U.K. corporate tax rate is usually between 20...
-
Jason Trevor owns a commercial bakery in Blakely, Georgia, that produces a variety of goods sold in grocery stores. Trevor is required by law to perform internal tests on food produced at his plant...
-
A 64-year-old patient of a pharmacy will retire at age 65 and will be switching from her employer-sponsored plan to Medicare. She wonders if she will have drug coverage like she does with her current...
-
Puerto Rico enacted a law that required specific labels on cement sold in Puerto Rico and imposed fines for any violations of these requirements. The law prohibited the sale or distribution of cement...
-
A state legislature enacted a statute that required any motorcycle operator or passenger on the states highways to wear a protective helmet. Jim Alderman, a licensed motorcycle operator, sued the...
-
At Ï = 1 rad/s find the transmission parameters of the network in Fig. 19.127 using PSpice. 1 H 1 H 1 F 1 F
-
Illini Company, Inc. Balance Sheet as of 12/31/20X0 Assets Current Assets: Cash $1,500,000 Accounts receivable, net 18,000 Inventory 50,000 Total current assets 1,568,000 Equipment 90,000 Goodwill...
-
The percent by mass of nitrogen for a compound is found to be 46.7%. Which of the following could be this species? N O
-
Complete the following table. Mass of Sample 4.24 g CH Moles of Sample 0.224 mol HO Molecules in Sample 2.71 X 1022 molecules CO Total Atoms in Sample 3.35 X 1022 total atoms in CH3OH sample
-
What amount (moles) is represented by each of these samples? a. 150.0 g Fe 2 O 3 b. 10.0 mg NO 2 c. 1.5 10 16 molecules of BF 3
-
QUESTION 5) (pressure drop in the pipe) 8.2 Water at 5 oC (p=999.9 kg/m3 and =1.519x10-3 kg/ms) flows at an average speed of 0.9 m/s in a horizontal pipe of 0.3 cm inner diameter and 10 cm length....
-
The stress at a point is shown on the element in (Figure 1). Suppose that = 2 ksi and Txz = 6 ksi. Part A Figure x 0 xz Determine the principal stresses. Express your answers in kilopounds per...
-
0.1 kg/s of water is heated from an inlet temperature of 25 C by passing through a thick-walled tube having an inner and outer radius of 10 mm and 25 mm, respectively. The outer surface of the tube...

Study smarter with the SolutionInn App


