Determine the indices for the planes in the cubic unit cell shown in Figure 3-40. X 3412
Question:
Determine the indices for the planes in the cubic unit cell shown in Figure 3-40.
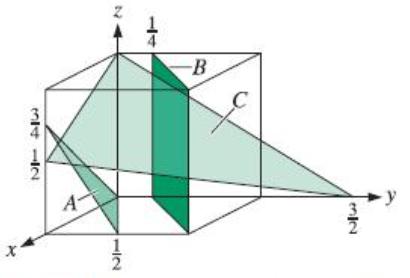
Transcribed Image Text:
X 3412 A 12 -B C 32
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (8 reviews)
The indices for the planes in the cubic unit cell shown in Figure 340 are 1...View the full answer

Answered By

Labindao Antoque
I graduated in 2018 with a Bachelor of Science degree in Psychology from Dalubhasaan ng Lungsod ng San Pablo. I tutored students in classes and out of classes. I use a variety of strategies to tutor students that include: lecture, discussions about the subject matter, problem solving examples using the principles of the subject matter being discussed in class , homework assignments that are directed towards reinforcing what we learn in class , and detailed practice problems help students to master a concept. I also do thorough research on Internet resources or textbooks so that I know what students need to learn in order to master what is being taught in class .
0.00
0 Reviews
10+ Question Solved
Related Book For 

The Science And Engineering Of Materials
ISBN: 9781305076761
7th Edition
Authors: Donald R. Askeland, Wendelin J. Wright
Question Posted:
Students also viewed these Engineering questions
-
Determine the indices for the planes in the cubic unit cell shown in Figure 3-39. C B N 113 A 72 y
-
Determine the indices for the directions shown in the following cubic unit cell: 3.32 Determine the indices for the directions shown in the following cubic unit cell: Crystallographic Planes 3.38 (a)...
-
Determine the Miller indices for the directions in the cubic unit cell shown in Figure 3-33.
-
By a work sampling study: Observed Data The machine is being used Proportion of manual labour (machine is running and/or (servicing time) T-% machine is random serviced) Allowance Time Proportion of...
-
This year, Yung Tseng, a U.S. citizen, supported his nephew who is attending school in the United States. Yung is a U.S. citizen, but his nephew is a citizen of Hong Kong. The nephew has a student...
-
Agassi Company uses a job order cost system in each of its three manufacturing departments. Manufacturing overhead is applied to jobs on the basis of direct labor cost in Department D, direct labor...
-
Describe various historical events that have led to the growth of hospitals and at times impeded their progress over the centuries.
-
Using the information from RE3-7, prepare Gold fingers closing entries for the current year. In RE3-7 Gold finger Corporation had account balances at the end of the current year as follows: sales...
-
Nora Incorporated sells a single product for $22. Variable costs include $8.58 for each unit plus a 8% sales commission. Fixed costs are $155,290 per month. Required: a. What is the contribution...
-
Pathfinder College is a small liberal arts college that wants to improve its admissions process. In particular, too many of its incoming freshmen have failed to graduate for a variety of reasons,...
-
From the information in this chapter, calculate the volume change that will occur when the cubic form of zirconia transforms into a tetragonal form.
-
Calculate the theoretical density of the three polymorphs of zirconia. The lattice constants for the monoclinic form are a = 5.156, b = 5.191, and c = 5.304 , respectively. The angle b for the...
-
List some characteristic properties that distinguish the metallic elements from the nonmetallic elements.
-
Why is Code Sec. 338 of limited interest today?
-
Which rival companies appear to be employing a broad low-cost strategy?
-
Barton Industries has operating income for the year of $3,500,000 and a 25% tax rate. Its total invested capital is $20,000,000 and its after-tax percentage cost of capital is 8%. What is the firms...
-
How does an acquiring corporation make a Code Sec. 338 election?
-
Patterson Brothers recently reported an EBITDA of $7.5 million and net income of $2.625 million. It had $1.5 million of interest expense, and its corporate tax rate was 25%. What was its charge for...
-
Suppose you are given the following data: ¢ Risk-free yearly interest rate is r = 6%. ¢ The stock price follows: St St1 = μSt + ÏStεt where the...
-
Without solving, determine the character of the solutions of each equation in the complex number system. 3x 2 3x + 4 = 0
-
Gallium has an orthorhombic structure with a0 = 0.45258 nm, b0 = 0.45186 nm, and c0 = 0.76570 nm. The atomic radius is 0.1218 nm. The density is 5.904 g/cm3, and the atomic weight is 69.72 g/mol....
-
Beryllium has a hexagonal crystal structure with a0 = 0.22858 nm and c0 = 0.35842 nm. The atomic radius is 0.1143 nm, the density is 1.848 g/cm3, and the atomic weight is 9.01 g/mol. Determine (a)...
-
Gold has 5.82 108 vacancies/cm3 at equilibrium at 300 K. What fraction of the atomic sites is vacant at 600 K?
-
differences between life cycle costing and traditional management accounting technique
-
How do relationships change with age? Might Sternberg's triangular theory of love differ between an 18-year-old couple and a 50-year-old couple? Provide examples.
-
Following is data for the available-for-sale securities held by Lindy Company as of December 31: calculate Name Number of Shares Cost per Share Fair Value per Share Total Cost Total Fair Value...

Study smarter with the SolutionInn App


