Determine the indices for the planes in the cubic unit cell shown in Figure 3-39. C B
Question:
Determine the indices for the planes in the cubic unit cell shown in Figure 3-39.
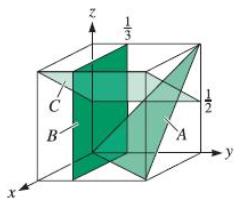
Transcribed Image Text:
C B N 113 A 72 y
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 54% (11 reviews)
The Crystal axes for the given cubic cell are 1 a 13 b 12 c which is given in the figure ...View the full answer

Answered By

User l_831638
Hello! My name is Sadhana.M.S. I made specilization in Nuclear Physics at Kuvempu university.I've been an physics tutor for degree level student . My motto is "teach student how to learn".
0.00
0 Reviews
10+ Question Solved
Related Book For 

The Science And Engineering Of Materials
ISBN: 9781305076761
7th Edition
Authors: Donald R. Askeland, Wendelin J. Wright
Question Posted:
Students also viewed these Engineering questions
-
Determine the indices for the planes in the cubic unit cell shown in Figure 3-40. X 3412 A 12 -B C 32
-
Determine the indices for the directions shown in the following cubic unit cell: 3.32 Determine the indices for the directions shown in the following cubic unit cell: Crystallographic Planes 3.38 (a)...
-
Determine the Miller indices for the directions in the cubic unit cell shown in Figure 3-33.
-
Solve the Given Requirements by using this Equation: Tangent Lines and Derivatives. x + cos y = 1 a) Find dy b) Evaluate at dx c) Draw the tangent line at 35 1 y = 0 -1 -2 05 1.5 d) Write the...
-
a. Who is liable for additional taxes on a joint return? b. Why is this so important?
-
Selecting Kanton Company's Financing Strategy and Unsecured Short-Term Borrowing Arrangement. Morton Mercado, the CFO of Kanton Company, carefully developed the estimates of the firm's total funds...
-
Discuss the types of discrimination that occur in the workplace.
-
For each of the following specific audit procedures, indicate the type of audit procedure it represents: (1) Inspection of records or documents, (2) Inspection of tangible assets, (3) Observation,...
-
Coors Company expects sales of $527,000 (6,200 units at $85 per unit). The company's total foxed costs are $264.000 and its versatile costs are $25 per unit. Compute (a) break-even in units and (b)...
-
You, CPA, work as an associate with Campbell and Associates LLP, a financial and business advisory firm. The board of directors of Pembroke Pulp and Paper Inc. (PPPI) has engaged your firm once again...
-
From the information in this chapter, calculate the volume change that will occur when the cubic form of zirconia transforms into a tetragonal form.
-
Calculate the theoretical density of the three polymorphs of zirconia. The lattice constants for the monoclinic form are a = 5.156, b = 5.191, and c = 5.304 , respectively. The angle b for the...
-
The trial balance as at 30 April 2014 of Timber Products Limited was balanced by the inclusion of the following debit balance: Difference on trial balance suspense account 2,513. Subsequent...
-
Describe the attributes of an attractive industry for a new venture.
-
Explain the tax status of child support payments.
-
In what sense do these market value ratios reflect investors opinions about a stocks risk and expected future growth?
-
What is a business plan? What are the advantages of preparing a business plan for a new venture? Explain your answer.
-
Generally, what is "at risk"?
-
You are given the following information concerning a stock denoted by St. Current value = 102. Annual volatility = 30%. You are also given the spot rate r = 5%, which is known to be constant...
-
Proposals have been made to ?sail? spacecraft to the outer solar system using the pressure of sunlight, or even to propel interstellar spacecraft with high-powered, Earth-based lasers. Sailing...
-
Tin atoms are introduced into an FCC copper crystal, producing an alloy with a lattice parameter of 3.7589 10-8 cm and a density of 8.772 g/cm3. Calculate the atomic percentage of tin present in the...
-
We replace 7.5 atomic percent of the chromium atoms in its BCC crystal with tantalum. X-ray diffraction shows that the lattice parameter is 0.29158 nm. Calculate the density of the alloy.
-
Suppose we introduce one carbon atom for every 100 iron atoms in an interstitial position in BCC iron, giving a lattice parameter of 0.2867 nm. For the Fe-C alloy, find the density and the packing...
-
How do metacommunity dynamics, encompassing processes of dispersal, colonization, and local extinction, influence species distributions and community structure across spatial scales, and what are the...
-
Assume the net assets transferred from the investee qualify as a business, as that term is defined in FASB ASC Master Glossary. At what amount will the Licenses be reported in the financial...
-
Determining how transactions change an accounting equation Marion Cassidy operates a service business called Cassidy Company. Cassidy Company uses the accounts shown in the following accounting...

Study smarter with the SolutionInn App


