Determine the phases that are present and the compositions for each phase in Cu-85 wt% Ag at
Question:
Determine the phases that are present and the compositions for each phase in Cu-85 wt% Ag at 800°C.
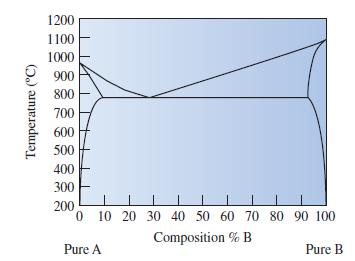
Transcribed Image Text:
Temperature (°C) 1200 1100 1000 900 800 700 600 500 400 300 200 0 10 20 30 40 50 60 70 80 90 100 Composition % B Pure A Pure B
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 69% (13 reviews)
ANSWER At 800 C the phases present are liquid an...View the full answer

Answered By

Aketch Cindy Sunday
I am a certified tutor with over two years of experience tutoring . I have a passion for helping students learn and grow, and I firmly believe that every student has the potential to be successful. I have a wide range of experience working with students of all ages and abilities, and I am confident that I can help students succeed in school.
I have experience working with students who have a wide range of abilities. I have also worked with gifted and talented students, and I am familiar with a variety of enrichment and acceleration strategies.
I am a patient and supportive tutor who is dedicated to helping my students reach their full potential. Thank you for your time and consideration.
0.00
0 Reviews
10+ Question Solved
Related Book For 

The Science And Engineering Of Materials
ISBN: 9781305076761
7th Edition
Authors: Donald R. Askeland, Wendelin J. Wright
Question Posted:
Students also viewed these Engineering questions
-
Determine the phases that are present and the compositions for each phase in Cu-55 wt% Ag at 600C. Data Form Problem 11-23 Consider a Pb-70% Sn alloy. Determine (a) If the alloy is hypoeutectic or...
-
Cite the phases that are present and the phase compositions for the following alloys: (a) 90 wt% Zn-10 wt% Cu at 400(C (750(F) (b) 75 wt% Sn-25 wt% Pb at 175(C (345(F) (c) 55 wt% Ag-45 wt% Cu at...
-
Cite the phases that are present and the phase compositions for the following alloys: (a) 90 wt% Zn-10 wt% Cu at 400C (750F) (b) 75 wt% Sn-25 wt% Pb at 175C (345F) (c) 55 wt% Ag-45 wt% Cu at 900C...
-
Consider a static (one-period), closed economy with one representative consumer, one rep- resentative firm, and a government. The level of capital K and government expenditures G in the economy are...
-
For the given of Problem 11, indicate: (a) How much would an interest arbitrageur earn if the foreign currency were at a forward premium of 1 percent per year? (b) What would happen if the foreign...
-
Lanning Company sells 160,000 units at $45 per unit. Variable costs are $27 per unit, and fixed costs are $975,000. Determine (A) The contribution margin ratio, (B) The unit contribution margin , (C)...
-
Is there a best method for completing performance appraisals for each of the three different types of employees in an MNE? If so, describe the ways in which an HRIS may help.
-
On July 31, 2019, after one month of operation, the general ledger of Michael Domenici, Consultant, contained the accounts and balances given below. INSTRUCTIONS 1. Prepare a partial worksheet with...
-
Kuala Lumpur Kepong Berhad stock currently sells for RM21.40 per share. The next expected annual dividend is RM0.80, and the growth rate is 4%. a. Find the expected rate of return on this stock. b....
-
Imperial Electronics Ltd. is a publicly owned company with 100,000 common shares outstanding. At the last executive committee meeting, Sandra Redgrave, CEO of the company, informed the board members...
-
A Pb-Sn alloy contains 45% and 55% at 100C. Determine the composition of the alloy. Is the alloy hypoeutectic or hypereutectic?
-
A Pb-Sn alloy contains 23% primary a and 77% eutectic microconstituent immediately after the eutectic reaction has been completed. Determine the composition of the alloy.
-
Identify the three cost elements that determine the cost of making a product (for external reporting).
-
It is July 2016. A mining company has just discovered a small deposit of gold. It will take six months to construct the mine. The gold will then be extracted on a more or less continuous basis for...
-
What is arbitrage? Explain the arbitrage opportunity when the price of a dually listed mining company stock is $50 (USD) on the New York Stock Exchange and $60 (CAD) on the Toronto Stock Exchange....
-
Suppose that on October 24, 2016, a company sells one April 2017 live-cattle futures contract. It closes out its position on January 21, 2017. The futures price (per pound) is 151.20 cents when it...
-
A trader buys two July futures contracts on orange juice. Each contract is for the delivery of 15,000 pounds. The current futures price is 120 cents per pound, the initial margin is $6,000 per...
-
One orange juice futures contract is on 15,000 pounds of frozen concentrate. Suppose that in September 2016 a company sells a March 2018 orange juice futures contract for 120 cents per pound. In...
-
The voltage across a 4-μF capacitor is shown in Fig. 6.45. Find the current waveform. v (V) 10 t (ms) -10
-
Interest Compounded Annually. When P dollars is invested at interest rate i, compounded annually, for t years, the investment grows to A dollars, where A = P(1 + i) t . Trevor's parents deposit $7800...
-
(a) Using only K sp from Table 6-3, calculate how many moles of Ca(OH) 2 will dissolve in 1.00 L of water. (b) How will the solubility calculated in part (a) be affected by the K 1 reaction in Table...
-
From the following equilibrium constants, calculate the equilibrium constant for the reaction HO,CCO,H 2H* + C,0.
-
Assuming complete dissociation of the salts, calculate the ionic strength of (a) 0.2 mM KNO 3 ; (b) 0.2 mM Cs 2 CrO 4 ; (c) 0.2 mM MgCl 2 plus 0.3 mM AlCl 3 .
-
What we did in the Portfolio beta.R file, but use another five different companie?
-
A worker who holds all of his wealth in his company's stock is in danger not spreading risk. hedging risk. "blowing up." innovating?
-
What factor should you consider when choosing the best investment for you? When you will need to use the money What your financial goals are How much risk you are willing to take All of these are...

Study smarter with the SolutionInn App


