How much of a temperature change is required to bring liquid magnesium from solid-liquid equilibrium to vaporliquid
Question:
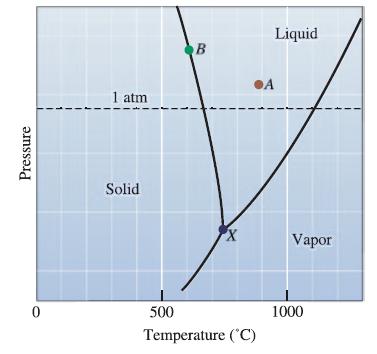
How much of a temperature change is required to bring liquid magnesium from solid-liquid equilibrium to vaporliquid equilibrium at 1 atmosphere?
Transcribed Image Text:
Pressure 0 1 atm Solid B X 500 Temperature (°C) Liquid A Vapor 1000
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 69% (13 reviews)
Temperature change Power Tempe...View the full answer

Answered By

Willis Omondi
Hi, I'm Willis Omondi, a proficient and professional academic writer. I have been providing high-quality content that best suits my clients and completing their work within the deadline. All my work has been 100% plagiarism-free, according to research from my services, especially in arts subjects and many others
0.00
0 Reviews
10+ Question Solved
Related Book For 

The Science And Engineering Of Materials
ISBN: 9781305076761
7th Edition
Authors: Donald R. Askeland, Wendelin J. Wright
Question Posted:
Students also viewed these Engineering questions
-
How should a monopsonist decide how much of a product to buy? Will it buy more or less than a competitive buyer? Explain briefly.
-
How much of a price premium do you think national brands ought to command over private brands? Justify your position.
-
A government is trying to decide how much of a public good to provide. The willingness-to-pay curves for each of its two citizens are as given in the diagram. The marginal cost curve for the public...
-
You are the manager of a local coffee shop. There are two types of customers in your market, coffee addicts, and casual drinkers. Because coffee addicts buy large amounts of coffee, they are more...
-
Starting with the exchange rate of R = $2/1, draw a figure showing the exchange rate under a crawling peg system with the nation appreciating its currency by 1 percent at the end of each month for...
-
The following are the eight balance-related audit objectives, seven tests of details of balances for accounts receivable, and seven tests of controls or substantive tests of transactions for the...
-
Briefly describe what a credit default swap is and how it is generally used.
-
A made a contract to construct a house for B. Subsequently, B sued A for breach of contract. A raised the defense that the contract was not binding because it was not sealed. Is this a valid defense?...
-
Superior Micro Products uses the weighted-average method in its process costing system. Data for the Assembly Department for May appear below: Materials Work in process, May 1 Cost added during May $...
-
As shown in Fig. 2, a "very long" tube A (inner tube made of aluminium) has been thermally shrink-fitted into another "very long" tube B (outer tube made of mild steel). The final dimensions of the...
-
How can solid solutions form in ceramic systems?
-
Do we need 100% solid solubility to form a solid solution of one material in another?
-
In industry, the relationship between wages and the quit ratio of employees is defined to be the percentage of employees that quit within 1 year of employment. The quit ratio of a large restaurant...
-
Floor heating is used to keep a room at a toasty 25C. On a cold day, the room loses heat through the walls, ceilings, door, and windows at the rate of 2,000 W. The heated floor area is 30 m 2 . The...
-
An insulated cardboard box is to be designed to transport a labile vaccine product that has to be kept at 78C in areas where lowtemperature refrigeration is not widely available. The box is a cube...
-
The design of an astronauts glove requires that you select the thickness of insulation needed to keep the skin surface temperature at 25C for long periods of time when the outer surface of the...
-
Serpe has reported on the development of smart windows, where the gap between two glass panes is filled with an aqueous solution containing hydrogel particles made of poly(#1- isopropylacrylamide) or...
-
A cube, 1.0-in. long on each side, is constructed of equal-sized, alternating parallel layers of copper and Pyrex glass. There are five layers of copper and five layers of glass. A question has...
-
Find vo in the circuit of Fig. 7.112 when vs = 6u(t). Assume that vo (0) = 1 V. 20 k02 10 k2 40
-
How has the globalization of firms affected the diversity of their employees? Why has increased diversity put an additional burden on accounting systems?
-
Which of the following statement(s) is(are) true? a. A radioactive nuclide that decays from 2.00 10 21 atoms to 5.0 10 20 atoms in 16 minutes has a half-life of 8.0 minutes. b. Nuclides with large...
-
A certain radioactive nuclide has a half-life of 3.00 hours. a. Calculate the rate constant in s -1 for this nuclide. b. Calculate the decay rate in decays/s for 1.000 mole of this nuclide.
-
Complete the following table with the nuclear particle that is produced in each nuclear reaction. Initial Nuclide 23 Pu 94 214Pb 82 60, 27 99- 43 Tc 93Np Product Nuclide 2351 92 214p 60 Ni 28 44Ru...
-
You and your partner own Gaia's Organic Grocery: You buy your romaine lettuce from Naturally Yours Garden Farm. Unknown to you, Naturally Yours' romaine has been infected by a microscopic parasite...
-
discuss the value drivers and sources of value, i.e. outputs/outcomes of the procurement and contracting operations, strategy, and tactics, or use of specific techniques. How did this way of...
-
Is there any operational or logistical connection between original value drivers (from the first acquisition perspective) and new value drivers (of the second acquisition perspective)? If yes, is...

Study smarter with the SolutionInn App


