Iron oxide ores, commonly a mixture of FeO and Fe 2 O 3 , are given the
Question:
Iron oxide ores, commonly a mixture of FeO and Fe2O3, are given the general formula Fe3O4. They yield elemental iron when heated to a very high temperature with either carbon monoxide or elemental hydrogen. Balance the following equations for these processes:
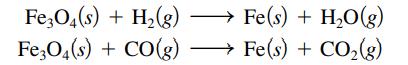
Transcribed Image Text:
Fe3O4(s) + H₂(g) Fe3O4(s) + CO(g) Fe(s) + H₂O(g) Fe(s) + CO₂(g)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 70% (10 reviews)
Fe3O4s 4H2g Fes 4H2Og Fe3O4s 3COg Fes 3CO2g The above equations are balanced ...View the full answer

Answered By

Labindao Antoque
I graduated in 2018 with a Bachelor of Science degree in Psychology from Dalubhasaan ng Lungsod ng San Pablo. I tutored students in classes and out of classes. I use a variety of strategies to tutor students that include: lecture, discussions about the subject matter, problem solving examples using the principles of the subject matter being discussed in class , homework assignments that are directed towards reinforcing what we learn in class , and detailed practice problems help students to master a concept. I also do thorough research on Internet resources or textbooks so that I know what students need to learn in order to master what is being taught in class .
0.00
0 Reviews
10+ Question Solved
Related Book For 

Chemistry
ISBN: 9781305957404
10th Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Question Posted:
Students also viewed these Engineering questions
-
As a body is heated to a very high temperature and becomes self luminous, the apparent color of the emitted radiation shifts from red to yellow and finally to blue as the temperature increases. Why...
-
Moist air is heated to a very high temperature. If the equilibrium composition consists of H2O, O2, N2, OH, H2, and NO, the number of equilibrium constant relations needed to determine the...
-
A mixture of Fe2O3 and FeO was found to contain 72.00% Fe by mass. What is the mass of Fe2O3 in 0.500 g of this mixture?
-
Give a recursive algorithm for finding the reversal of a bit string. (See the definition of the reversal of a bit string in the preamble of Exercise 34 in Section 5.3.)
-
Tim retired during the current year at age 58. He purchased an annuity from American National Life Company for $40,000. The annuity pays Tim $500 per month for life. a. Compute Tims annual exclusion....
-
Henkel Company is considering three long-term capital investment proposals. Each investment has a useful life of 5 years. Relevant data on each project are as follows. Depreciation is computed by the...
-
Explain how consent differs among competent patients, minors, guardians, and incompetent patients.
-
Minott Jewelers, Ltd., purchased store fixtures, display cases, and a maximum-security commercial safe for a lump-sum price of $14,000 from a bankrupt competitor. Appraised values were as follows:...
-
Given the following Venn diagram, find n[(CUB)]. 11 10 10 15 15 6 12 5 11 B U
-
Penang Electronics (PE) is a contract manufacturer that produces and packages private label products for various retail chains, including Target, Best Buy, Staples, and Office Max. In each case, the...
-
Balance each of the following chemical equations. a. KO (s) + HO(1)KOH(aq) + O(g) + HO(aq) b. FeO3(s) + HNO3(aq) Fe(NO3)3(aq) + HO(1) c. NH3(g) + O(g) NO(g) + HO(g) d. PC1,(1) + HO(1) H3PO4(aq) +...
-
A common demonstration in chemistry courses involves adding a tiny speck of manganese(IV) oxide to a concentrated hydrogen peroxide (H 2 O 2 ) solution. Hydrogen peroxide decomposes quite...
-
From the ten books that youve recently bought but not read, you plan to take four with you on vacation. How many different sets of four books can you take?
-
Assume that today is December 31, 2021, and that the following information applies to Abner Airlines: After-tax operating income [EBIT(1 2 T)] for 2022 is expected to be $400 million. The...
-
Adamson Corporation is considering four average-risk projects with the following costs and rates of return: The company estimates that it can issue debt at a rate of r d = 10%, and its tax rate is...
-
What condition regarding cash flows would cause more than one IRR to exist? Project MM has the following cash flows: 0 - $1,000 End-of-Year Cash Flows 1 2 $2,000 $2,000 3 - $3,350
-
A firm with a 14% WACC is evaluating two projects for this years capital budget. After-tax cash flows are as follows: a. Calculate NPV, IRR, MIRR, payback, and discounted payback for each project. b....
-
Components Manufacturing Corporation (CMC) has an all-common-equity capital structure. It has 200,000 shares of $2 par value common stock outstanding. When CMCs founder, who was also its research...
-
In this exercise we work with the Black- Scholes setting applied to foreign currency denominated assets. We will see a different use of Girsanov theorem. [For more details see Musiela and Rutkowski...
-
Prove the result that the R 2 associated with a restricted least squares estimator is never larger than that associated with the unrestricted least squares estimator. Conclude that imposing...
-
The mobility of a u- ion in aqueous solution is 4.01 x 10-8 m2 S-1 V-1 at 25C. The potential difference between two electrodes placed in the solution is 12.0 V. If the electrodes are 1.00 cm apart,...
-
What fraction of the total current is carried by er when current flows through an aqueous solution of NaCI at 25C?
-
What fraction of the total current is carried by er when current flows through an aqueous solution of NaCI at 25C?
-
1. Your organization has corporate-owned mobile devices. You are creating security controls to find these devices if they are lost or stolen. Which of the following BEST meets this goal? A. ...
-
In each box of cereal that it produces, Snoopy Cereal Corp. includes a special coupon.The purchaser may redeem 10 coupons for a cheese grater (premium).Each grater costsSnoopy $0.90. During2017,...
-
Are the payoffs identical for an investor who buys a call option and sells a put option? Explain.

Study smarter with the SolutionInn App


