Of the metals listed in Table 22-1, which have the highest and lowest specific heats? Also list
Question:
Of the metals listed in Table 22-1, which have the highest and lowest specific heats? Also list their atomic masses
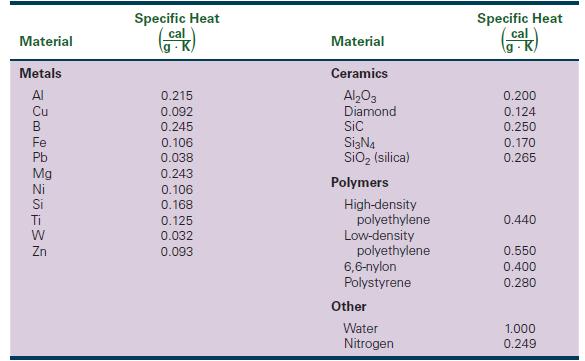
Transcribed Image Text:
Material Metals Al Cu B Fe Pb Mg Ni Si Ti W Zn Specific Heat cal 0.215 0.092 0.245 0.106 0.038 0.243 0.106 0.168 0.125 0.032 0.093 Material Ceramics Al₂O3 Diamond Sic Si3N4 SiO₂ (silica) Polymers High-density polyethylene Low-density polyethylene 6,6-nylon Polystyrene Other Water Nitrogen Specific Heat cal K 0.200 0.124 0.250 0.170 0.265 0.440 0.550 0.400 0.280 1.000 0.249
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (5 reviews)
ANSWER From the table we can see that the metal with the highest specific heat is magnesium Mg ...View the full answer

Answered By

User l_917591
As a Business Management graduate from Moi University, I had the opportunity to work as a tutor for undergraduate students in the same field. This experience allowed me to apply the theoretical knowledge I had gained in a practical setting, while also honing my teaching and communication skills.
As a tutor, I was responsible for conducting tutorial sessions, grading assignments and exams, and providing feedback and support to my students. I also assisted with the preparation of course materials and collaborated with other tutors and professors to ensure consistency in teaching and assessment.
0.00
0 Reviews
10+ Question Solved
Related Book For 

The Science And Engineering Of Materials
ISBN: 9781305076761
7th Edition
Authors: Donald R. Askeland, Wendelin J. Wright
Question Posted:
Students also viewed these Engineering questions
-
Of the metals listed in Table 19-1, which are the most and least conductive? Material Superconductors Hg, Nb3Sn YBaCu3O7-x MgB Metals Alkali metals Na K Alkali earth metals Mg Ca Group 3B metals Al...
-
In Figure 20-9(b), what materials have the highest and lowest saturation magnetizations and coercivities? Square loop for computer applications Inductance (a) Soft magnet for electrical applications...
-
For each of the metals listed in the following table, compute the Pilling-Bedworth ratio. Also, on the basis of this value, specify whether you would expect the oxide scale that forms on the surface...
-
Which of the following are valid in a Java file, listed in the order in which they are declared? (Choose two.) A. A package-private class declaration and a public interface declaration B. Two package...
-
In the business world of the 21st Century, will it be possible to make critical marketing decisions without marketing research? Why or why not?
-
Given that f is a differentiable function with f (2, 5) = 6, f x (2, 5) = 1, and f y (2, 5) = -1, use a linear approximation to estimate f(2.2, 4.9).
-
Allan and Koraev both owned condominiums in the same building. Koraevs unit was directly above Allans. While Allan lived in her own unit, Koraev leased his. The leasing of Koraevs unit was managed by...
-
Schultz Electronics manufactures two large-screen television models: the Royale which sells for $1,600, and a new model, the Majestic, which sells for $1,300. The production cost computed per unit...
-
23. If an item retails for $320 after being marked up by 85%, what was the dollar amount of the cost?
-
Use the simple transistor model (with perfect saturation and no bulk effect) for this question. We want to compare the current ID drawn by a single nMOS transistor and ID2 drawn by two series...
-
Calculating Power in Decibels. In an optical communications system or electrical power transmission system, the power or signal often is transferred between several components. The decibel (dB) is a...
-
What is the minimum accelerating voltage required to produce Ka x-rays in nickel?
-
Lindstrom Company produces two fountain pen models. Information about its products follows: Lindstroms fixed costs total $78,500. Required: 1. Determine Lindstroms weighted-average unit contribution...
-
4. Suppose Michelle's tastes for haircuts (x1) and spending on other goods (x2) per year can be represented by the separable utility function u(x1, x2)=600x1" + 2x2. (a) Derive an expression for...
-
A rifle with a weight of 35 N fires a 5.5-g bullet with a speed of 220 m/s. (a) Find the recoil speed of the rifle. 0.339 m/s (b) If a 650-N man holds the rifle firmly against his shoulder, find the...
-
Define the following: (a) Audit instrument (b) Field notes (c) Life cycle of documents (d) Audit scope (e) Audit fatigue (f) Audit trail (g) Compliance
-
What is FEMA and who does the FEMA director report to? How does FEMA coordinate with state and local agencies? We see FEMA respond to major weather events like Hurricane Katrina, but what are some...
-
When an employer has reason to believe a worker who was already trained in fall protection does not fully understand the safeguards involved in the training, by Code what must the employer do?
-
For the scatterplots and graphs in Exercises 71-76, determine which, if any, of the following functions might be used as a model for the data. a) Linear, f(x) = mx + b b) Quadratic, f(x) = ax2 + bx +...
-
In order to get an idea on current buying trends, a real estate agent collects data on 10 recent house sales in the area. Specifically, she notes the number of bedrooms in each house as follows: a....
-
As quality control engineer for your company, you must approve all material shipments from your suppliers. Part of this job involves testing random samples from each delivery and making sure they...
-
When estimating the live load for a new bridge design, you want that estimate to be conservative. In other words, you want to error on the safe side by basing the estimate on the worst possible...
-
Determine the forces acting on the ends of the rope by (1) drawing the free-body diagram, (2) counting unknowns and equations to check determinacy, (3) writing the equilibrium equations, and (4)...
-
search box near the bottom of the screen. In the search box, instruct it to draft a contract between RJ Hunt, seller, and DJ March, buyer, for the sale of a house at 123 Jackson St., Macomb, IL...
-
On the Customers worksheet, use TEXTJOIN to return Wilbur's first and last name, separated by a space, to D4. Then copy the formula to D5:D13
-
Hot Property Got to Be the Shoes - NIKE He rises into the air with his legs flared and a basketball held high. It's hard to forget the slam- dunk image of Michael Jordan - especially when it's...

Study smarter with the SolutionInn App


