Sketch the galvanic cells based on the following overall reactions. Show the direction of electron flow, the
Question:
Sketch the galvanic cells based on the following overall reactions. Show the direction of electron flow, the direction of ion migration through the salt bridge, and identify the cathode and anode. Give the overall balanced equation. Assume that all concentrations are 1.0 M and that all partial pressures are 1.0 atm.
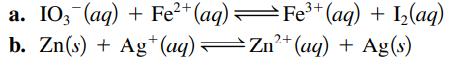
Transcribed Image Text:
2+ a. IO3(aq) + Fe²+ (aq) — Fe³+ (aq) + 1₂(aq) = b. Zn(s) + Ag+ (aq) Zn²+ (aq) + Ag(s)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (10 reviews)
ANSWER a The galvanic cell for the specified overall re...View the full answer

Answered By

Aketch Cindy Sunday
I am a certified tutor with over two years of experience tutoring . I have a passion for helping students learn and grow, and I firmly believe that every student has the potential to be successful. I have a wide range of experience working with students of all ages and abilities, and I am confident that I can help students succeed in school.
I have experience working with students who have a wide range of abilities. I have also worked with gifted and talented students, and I am familiar with a variety of enrichment and acceleration strategies.
I am a patient and supportive tutor who is dedicated to helping my students reach their full potential. Thank you for your time and consideration.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Chemistry
ISBN: 9781305957404
10th Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Question Posted:
Students also viewed these Engineering questions
-
Sketch the galvanic cells based on the following half-reactions. Calculate Ïo, show the direction of electron flow and the direction of ion migration through the salt bridge, identify the...
-
Sketch the galvanic cells based on the following overall reactions. Calculate o , show the direction of electron flow and the direction of ion migration through the salt bridge, identify the cathode...
-
Assume both electron and hole concentrations in a semiconductor are raised by n above their equilibrium values. Define a net minority carrier lifetime t by R = n/t. give expressions for t in terms of...
-
Which of the following statements about an acquiescence is correct? a. Acquiescences are published only for certain regular decisions of the U.S. Tax Court. b. The IRS does not issue acquiescences to...
-
Robert Shiller of Yale University has suggested a variation on ARMs in which mort-gage interest rates are tied to inflation, not to short-term interest rates. Discuss the pros and cons of this idea...
-
An employment information service claims that the standard deviation of the annual salaries for public relations managers is less in Florida than in Louisiana. You select a sample of public relations...
-
Match the measures of worth in the first column with one (or more) of the analysis approaches that is (are) appropriate for that measure. Measure of Worth (a) Annual Worth (b) External Rate of Return...
-
Golden Corp., a merchandiser, recently completed its 2013 operations. For the year, (1) all sales are credit sales, (2) all credits to Accounts Receivable reflect cash receipts from customers, (3)...
-
Briefly describe TWO (2) drawbacks of NoSQL in comparison with RDBMS ?
-
For this assignment, students need to identify the ASC (i.e., four numbers) that can serve as authoritative guidance for the issue raised in each of the cases below and provide a short response. 1....
-
Calculate G and K at 25 C for the reactions in Exercises 37 and 41. Data in Exercise 37 Sketch the galvanic cells based on the following overall reactions. Show the direction of electron flow, and...
-
Which of the following statements concerning corrosion is(are) true? For the false statements, correct them. a. Corrosion is an example of an electrolytic process. b. Corrosion of steel involves the...
-
True or false: A dynamic memory will hold its data as long as electrical power is applied.
-
How will you react to the resorts reputation of rough starts, but it gets better after you've been working there for a while?
-
How does critically analyzing diversity add value to how you interact with people and understand social or global issues/events?
-
What is infinite loading and how does it impact Capacity Requirements Planning (CRP) and MRP? How is Distribution Requirements Planning (DRP) used to enhance supply chains?
-
What do we mean by "sensitivity analysis"? What two basic questions can we answer using sensitivity analysis?
-
Discuss the following terms along with examples Upward delegation. Sideward delegation. Downward delegation.
-
Repeat problem 8-1-4 [OZE] assuming a compressor efficiency of 80% and a turbine efficiency of 90%, and determine (a) The turbine exit temperature. Use the PG model. (b) Back work ratio, (c) The...
-
(a) Find the equation of the tangent line to f(x) = x 3 at the point where x = 2. (b) Graph the tangent line and the function on the same axes. If the tangent line is used to estimate values of the...
-
If an electron passes through an electrical potential difference of 1 V, it has an energy of 1 electron-volt. What potential difference must it pass through in order to have a wavelength of 0.300 nm?
-
Calculate the longest and the shortest wavelength observed in the Balmer series.
-
X-rays can be generated by accelerating electrons in a vacuum and letting them impact on atoms in a metal surface. If the 1250. eV kinetic energy of the electrons is completely converted to the...
-
what will happen to Miami, Florida if the temperature increase to 1.5C.
-
On March 31, 2021, a farmer knows he will need to sell 15,000 bushels of wheat some time in October 2021. The November wheat futures price is 778.3 cents per bushel. Each wheat futures contract is on...
-
If you happen to be looking at the fi lament of an incandescent light bulb just before it burns out, you might notice that the fi lament glows brighter just before it burns out. Explain these...

Study smarter with the SolutionInn App


