Sulfur trioxide, SO 3 , is produced in enormous quantities each year for use in the synthesis
Question:
Sulfur trioxide, SO3, is produced in enormous quantities each year for use in the synthesis of sulfuric acid.
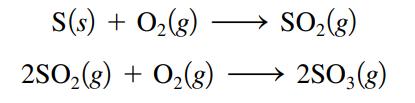
What volume of O2(g) at 350.οC and a pressure of 5.25 atm is needed to completely convert 5.00 g sulfur to sulfur trioxide?
Transcribed Image Text:
S(s) + 0₂ (8) + O₂(g) 2SO₂(g) SO₂(g) 2SO3(g)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 55% (9 reviews)
The volume 500 g x 1 mol SO38409 g SO3 20 moles O21 mol O2 0...View the full answer

Answered By

Brian Otieno
I'm Brian , an experienced professional freelancer with countless hours of success in freelancing many subjects in different disciplines. Specifically, I have handled many subjects and excelled in many disciplines. I have worked on many Computer Science projects and have been able to achieve a lot in that field. Additionally, I have handled other disciplines like History, Humanities, Social Sciences, Political science, Health care and life science, and Religion / Theology. My experience generally in these subjects has made me able to deliver high-quality projects in a very timely fashion. I am very reliable at my job and will get the work done in time, no matter what. In Addition, I have managed to ensure that the work meets my client's expectations and does not cause an error. I am a hard-working and diligent person who is highly responsible for everything I do. Generally, Freelancing has made me more accountable for doing my job. Additionally, I have had a passion for writing for the last seven years in this field.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Chemistry
ISBN: 9781305957404
10th Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Question Posted:
Students also viewed these Engineering questions
-
An ideal gas at 15.5 C and a pressure of 1.72 105 Pa occupies a volume of 2.81 m3. (a) How many moles of gas are present? (b) If the volume is raised to 4.16 m3 and the temperature raised to 28.2...
-
Air at a temperature of 30 C and a pressure of 100 kPa has a relative humidity of 80%. Calculate (a) The molal humidity of the air (b) Saturated molal humidity of this air if its temperature is...
-
5 liters of air with a humidity of 0.6, a temperature of 20 C and a pressure of 75 cm Hg it is pressed together to 2 liters at a temperature of 25 C. Calculate all missing state parameters, the...
-
Write a function my_ieee_2_dec(ieee), where icce is a string contains 64 char- acters of ones and zeros representing a 64-bit IEEE754 number. The output should be d, the equivalent decimal...
-
Adriana Santosh is in the business of selling antique furniture. As her company has increased significantly the Internet portion of their business, Adriana has determined that she does not need as...
-
Find the first and second derivatives of the function. G(r) = \F + r
-
Using the AISC equations, select from Appendix B the lightest-weight wide-flange A992 steel column that is \(12 \mathrm{ft}\) long and supports an axial load of 20 kip. The ends are pinned.
-
ADJUSTING ENTRIES INCLUDING ADJUSTMENT FOR UNDERAPPLIED/ OVERAPPLIED FACTORY OVERHEAD Prepare the December 31 adjusting journal entries for Johnson Company. Data are as follows: (a) Factory overhead...
-
To match organization structure to strategy, organizations outsource part of their operations. What type of activities should organizations outsource? What should they not outsource? What is the goal...
-
In each of the following exercises, the assignment is to prepare an office memo. Each assignment contains an assignment memo from the supervising attorney that includes all the available facts of the...
-
A typical adult inhales 450 mL of air in any one breath. How many air particles are in a typical breath at 745 torr and 22C?
-
A 2.50-L container is filled with 175 g argon. a. If the pressure is 10.0 atm, what is the temperature? b. If the temperature is 225 K, what is the pressure?
-
A 17,000-kg jet takes off from an aircraft carrier via a catapult (Fig. 6-39a). The gases thrust out from the jet's engines exert a constant force of 130 kN on the jet; the force exerted on the jet...
-
(java) // create an array of 4 grocery items as Strings // create a LinkedList // add them one by one // use assertEquals to check they're added in correct order // insert a 5th grocery item (as a...
-
How to get the deadband between 2 PWM signals using Arduino Uno. The code below is how I generated 2 PWM signals. // Synchronize PWM on channels outputs OC1A, OC1B at 20Hz and OC3A at 40Hz void...
-
Write a project proposal on Predictive Policing Using Long Short Term Memory. The proposal must contain but not limted to; Abstract Introduction Project Problem Statement Objectives and Expectations...
-
A 0.2m drum of an immiscible liquid fuel, with weight of 1100lb, has spilled into a diked area and caught on fire. What is the specific gravity of the liquid fuel and if water is used to suppress the...
-
With the speed of computers today, should we even care about big O notation?
-
A linear transformation L: V W is said to map V onto W if L(V) = W. Show that the linear transformation L defined by L(x) = (x1.x1 +x2,x1 +.x2 + x3)T maps R3 onto R3.
-
a) Calculate the goodwill that was paid by Major Ltd on the acquisition of Minor Ltd. [10 marks] b) Prepare the consolidated statement of financial position for Major Ltd at 31 July 20X8. [30 marks]...
-
Using any compounds that contain two carbon atoms or fewer, show a way to prepare a racemic mixture of (2R, 3R)- 2,3-dihydroxypentane and (2S,3S)-2,3-dihydroxypentane.
-
For each pair of compounds below, identify the more acidic compound: (a) (b) (c) (d) (e) (f) (g) (h) SH
-
Paclitaxel (marketed under the trade name TaxolTM) is found in the bark of the Pacific yew tree, Taxus berevifolia, and is used in the treatment of cancer: (a) Draw the enantiomer of paclitaxel. (b)...
-
Magic Realm, Incorporated, has developed a new fantasy board game. The company sold 40,500 games last year at a selling price of $69 per game. Fixed expenses associated with the game total $729,000...
-
CALCULATE THE INITIAL COSTS, CALCULATE THE OPERATING CASH FLOW, CALCULATE THE TERMINAL NON OPERATING CASH FLOW, CALCULATE THE NPV . Initial Equipment $ 6 5 , 0 0 0 Project Life 3 Years Sales$ 5 5 ,...
-
A description of how each of the two management styles selected distinctively create organizational cultures. Describe the strengths and weaknesses of the two styles. Explain how these differences...

Study smarter with the SolutionInn App


