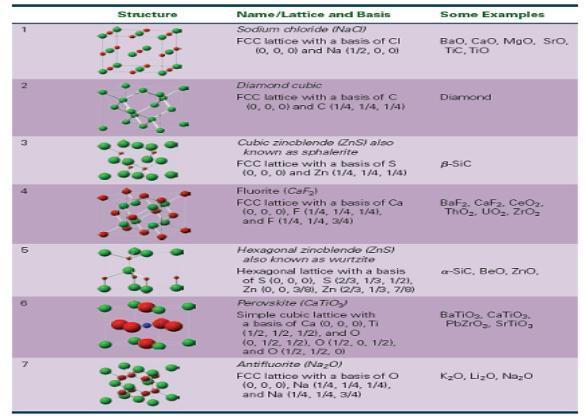
Table 15-4 describes the cubic zincblende structure as a face-centered cubic lattice with a basis of S
Question:
Table 15-4 describes the cubic zincblende structure as a face-centered cubic lattice with a basis of S (0, 0, 0) and Zn (1/4, 1/4, 1/4). Using S as one ion of the basis, give three other equivalent descriptions for the position of the Zn ion.
Transcribed Image Text:
Structure Name/Lattice and Basis Sodium chloride (NaC)) FCC lattice with a basis of Cl (0, 0, 0) and Na (1/2, 0, 01 Diamond cubic FCC lattice with a basis of C (0, 0, 0) and C (1/4, 1/4, 1/4) Cubic zincblende (ZnS) also known as sphalerite FCC lattice with a basis of S (0, 0, 0) and Zn (1/4, 1/4, 1/4) Fluorite (CaF₂ FCC lattice with a basis of Ca (0, 0, 0), F (1/4, 1/4, 1/4), and F (1/4, 1/4, 3/4) Hexagonal zincblende (ZnS) also known as wurtzite Hexagonal lattice with a basis of S (0, 0, 0), S (2/3, 1/3, 1/2), Zn (0, 0, 3/0), Zn (2/3, 1/3, 7/8) Perovskite (CaTiO₂) Simple cubic lattice with a basis of Ca (0, 0, 0), Ti (1/2, 1/2, 1/2), and O (0. 1/2, 1/2). O (1/2, 0. 1/2). and O (1/2, 1/2, 0) Antifluorite (Na₂O) FCC lattice with a basis of O (0, 0, 0), Na (1/4, 1/4, 1/4). and Na (1/4, 1/4, 3/4) Some Examples Bao, Cao. MaO, SIO, TIC, TIO Diamond B-Sic BaF2. CaF₂. CeO₂. ThO₂. UO₂. ZrO₂ a-SiC, BeO, ZnO, BaTiO₂, CaTiO₂. PbZrO₂. SrTiO₂ K₂O, Li₂O, Na₂O
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 44% (9 reviews)
Solution The three include i Assign each atom a Cartesian ...View the full answer

Answered By

James Warinda
Hi! I’m James Otieno and I'm an experienced professional online tutor with countless hours of success in tutoring many subjects in different disciplines. Specifically, I have handled general management and general business as a tutor in Chegg, Help in Homework and Trans tutor accounts.
I believe that my experience has made me the perfect tutor for students of all ages, so I'm confident I can help you too with finding the solution to your problems. In addition, my approach is compatible with most educational methods and philosophies which means it will be easy for you to find a way in which we can work on things together. In addition, my long experience in the educational field has allowed me to develop a unique approach that is both productive and enjoyable.
I have tutored in course hero for quite some time and was among the top tutors awarded having high helpful rates and reviews. In addition, I have also been lucky enough to be nominated a finalist for the 2nd annual course hero award and the best tutor of the month in may 2022.
I will make sure that any student of yours will have an amazing time at learning with me, because I really care about helping people achieve their goals so if you don't have any worries or concerns whatsoever you should place your trust on me and let me help you get every single thing that you're looking for and more.
In my experience, I have observed that students tend to reach their potential in academics very easily when they are tutored by someone who is extremely dedicated to their academic career not just as a businessman but as a human being in general.
I have successfully tutored many students from different grades and from all sorts of backgrounds, so I'm confident I can help anyone find the solution to their problems and achieve
0.00
0 Reviews
10+ Question Solved
Related Book For 

The Science And Engineering Of Materials
ISBN: 9781305076761
7th Edition
Authors: Donald R. Askeland, Wendelin J. Wright
Question Posted:
Students also viewed these Engineering questions
-
Table 15-4 describes the sodium chloride structure as a face-centered cubic lattice with a basis of Cl (0, 0, 0) and Na (1/2, 0, 0). Using Na (0, 0, 0) as one ion of the basis, give three equivalent...
-
Table 15-4 describes the sodium chloride structure as a face-centered cubic lattice with a basis of Cl (0, 0, 0) and Na (1/2, 0, 0). Using Cl (0, 0, 0) as one ion of the basis, give three equivalent...
-
Using Table 1.3, determine what charge the ion will carry when each of the following elements reacts to form an ionic compound: Al, Li, S, and O. Table 1.3 Table 1.3 Valence Electrons of the First 18...
-
Explain why b x = e x ln b .
-
A community health status survey obtained the following demographic information from the respondents: Compute the relative frequency and cumulative relative frequency of the age groups. Age 18 to 29...
-
The Walt Disney Company's annual report for the year ended October 3, 2015, includes income statements for the years ending in 2013, 2014, and 2015. Net income for these three years is as follows...
-
Consider the Hald cement data in Table B.21. a. From the matrix of correlations between the regressors, would you suspect that multicollinearity is present? b. Calculate the variance inflation...
-
How do the customer and contractor know if each one completely understands the statement of work, the work breakdown structure, and the program plan?
-
List the characteristics (seven) of insurance contracts and explain each characteristic of insurance contract. 4. List and briefly explain the types of insurers as classified by place of...
-
McLean, a former vice president of International Harvesters international division, was charged with violation of the Foreign Corrupt Practices Act (FCPA). Harvester pleaded guilty and admitted that...
-
If a face-centered cubic crystal structure has all of its tetrahedral sites filled with atoms that fit perfectly into each tetrahedral site, what is its atomic packing factor?
-
Convert the properties of Al 2 O 3 from Table 15-3 into values with SI units. Table 15-3 Mechanical properties of selected advanced ceramics Flexural Strength (psi) Material AlO3 SiC (sintered) Si3N4...
-
Assume that a system has a very large number of energy levels given by the formula l = 0 l 2 with 0 = 1.75 10 -22 J, where l takes on the integral values 1, 2, 3, . . Assume further that the...
-
What inputs are required for the dividend growth approach?
-
Which capital structure theories does the empirical evidence seem to support?
-
A computer sells for $1,500 U.S. dollars. In the spot market, $1 = 115 Japanese yen. If purchasing power parity holds, what should be the price (in yen) of the same computer in Japan?
-
Explain how asymmetric information and signals affect capital structure decisions.
-
What are three ways to estimate the expected dividend growth rate?
-
Draw the structure of an optically active triglyceride containing one equivalent of stearic acid and two equivalents of oleic acid. Draw the products expected when this triglyceride reacts with the...
-
In Problem 8.43, determine the smallest value of for which the rod will not fall out of the pipe. IA -3 in.-
-
An object starts with an initial velocity of 3 m/s at t = 0, and it accelerates with an acceleration of a(t) = 7t m/s 2 . Find the total distance the object travels in 4 s.
-
The equation for the voltage (t) across a capacitor as a function of time is where i(t) is the applied current and Q 0 is the initial charge. A certain capacitor initially holds no charge. Its...
-
A certain objects acceleration is given by a(t) = 7t sin 5t m/s 2 . Compute its velocity at t = 10 s if its initial velocity is zero.
-
. A person can be a product. Please give an example of this.
-
64- 56- 48- 40- 32- 24- PPE Neighbor 16- The graph to the right shows how many pounds of apples and pounds of cherries you and your neighbor can each pick in one week. For example, if you devote all...
-
Consider the table below. Compute for the GDP deflator and the two means of computing the inflation rate. You may use Excel but do place your answers in the table below. GDP (current prices) GDP...

Study smarter with the SolutionInn App


