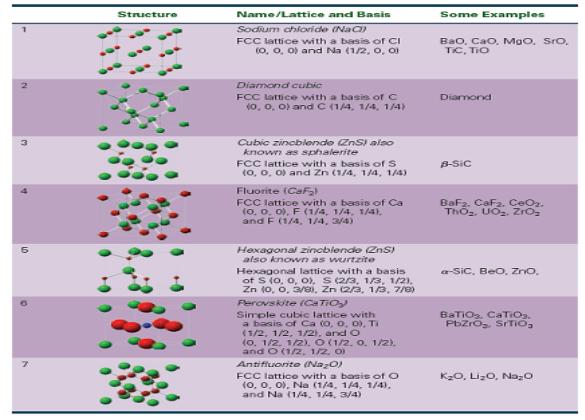
Table 15-4 describes the sodium chloride structure as a face-centered cubic lattice with a basis of Cl
Question:
Table 15-4 describes the sodium chloride structure as a face-centered cubic lattice with a basis of Cl (0, 0, 0) and Na (1/2, 0, 0). Using Cl (0, 0, 0) as one ion of the basis, give three equivalent descriptions for the position of the Na ion.
Transcribed Image Text:
Structure Name/Lattice and Basis Sodium chloride (NaC)) FCC lattice with a basis of Cl (0, 0, 0) and Na (1/2, 0, 01 Diamond cubic FCC lattice with a basis of C (0, 0, 0) and C (1/4, 1/4, 1/4) Cubic zincblende (ZnS) also known as sphalerite FCC lattice with a basis of S (0, 0, 0) and Zn (1/4, 1/4, 1/4) Fluorite (CaF₂ FCC lattice with a basis of Ca (0, 0, 0), F (1/4, 1/4, 1/4), and F (1/4, 1/4, 3/4) Hexagonal zincblende (ZnS) also known as wurtzite Hexagonal lattice with a basis of S (0, 0, 0), S (2/3, 1/3, 1/2), Zn (0, 0, 3/0), Zn (2/3, 1/3, 7/8) Perovskite (CaTiO₂) Simple cubic lattice with a basis of Ca (0, 0, 0), Ti (1/2, 1/2, 1/2), and O (0. 1/2, 1/2). O (1/2. 0. 1/2). and O (1/2, 1/2, 0) Antifluorite (Na₂O) FCC lattice with a basis of O (0, 0, 0), Na (1/4, 1/4, 1/4). and Na (1/4, 1/4, 3/4) Some Examples Bao, Cao. MaO, SIO, TIC, TIO Diamond B-Sic BaF2. CaF₂. CeO₂. ThO₂. UO₂. ZrO₂ a-SiC, BeO, ZnO, BaTiO₂, CaTiO₂. PbZrO₂. SrTiO₂ K₂O, Li₂O, Na₂O
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 61% (13 reviews)
The position of the Na ion can be described in three diffe...View the full answer

Answered By

Hande Dereli
Enthusiastic tutor, skilled in ACT and SAT tutoring. Raised one student's score on the SATs from 1100 combined to 1400. Graduated with a 3.9 GPA from Davidson College and led a popular peer tutoring group for three years. Scored in the top 0.06% in the nation on the SATs. The real reason I'm the one to help you nail the test? Results. Clients invariably praise my ability to listen and communicate in a low-stress, fun way. I think it's that great interaction that lets me raise retest SAT scores an average of 300 points.
0.00
0 Reviews
10+ Question Solved
Related Book For 

The Science And Engineering Of Materials
ISBN: 9781305076761
7th Edition
Authors: Donald R. Askeland, Wendelin J. Wright
Question Posted:
Students also viewed these Engineering questions
-
Table 15-4 describes the sodium chloride structure as a face-centered cubic lattice with a basis of Cl (0, 0, 0) and Na (1/2, 0, 0). Using Na (0, 0, 0) as one ion of the basis, give three equivalent...
-
Table 15-4 describes the cubic zincblende structure as a face-centered cubic lattice with a basis of S (0, 0, 0) and Zn (1/4, 1/4, 1/4). Using S as one ion of the basis, give three other equivalent...
-
Let Find x , y , xy , and yx , and state the domain for each partial derivative. f(x, y) = [y, y = 0 1-y, y < 0.
-
Use the COUNTIF function to construct a frequency distribution of the types of loans in the Excel file Credit Risk Data and develop a column chart to express the results visually.
-
Spencer Software Company has assets of $1,020,000 and liabilities of $552,000. a. Prepare the owners' equity section of the company's balance sheet under each of the following independent...
-
Table B. 14 presents data on the transient points of an electronic inverter. Use all possible regressions and the \(C_{p}\) criterion to find an appropriate regression model for these data....
-
Base your answers to the following questions on the financial statements for Leons Furniture imited/Meubles Lon Lte in Exhibits 1.27A to 1.27D. In the questions below, the year 2016 refers to Leons...
-
Business Law Assignment: 2.) Accidents on the workplace: methods of treatment. b.) In your research paper, explore how different businesses handle such situations. Are these methods always legal?
-
In the tax-plus-rebate example discussed in the text, suppose that the government adjusts the size of the rebate so that the consumer stays on her initial indifference curve (U2 in Figure 4.4). Show...
-
If a face-centered cubic crystal structure has all of its tetrahedral sites filled with atoms that fit perfectly into each tetrahedral site, what is its atomic packing factor?
-
Convert the properties of Al 2 O 3 from Table 15-3 into values with SI units. Table 15-3 Mechanical properties of selected advanced ceramics Flexural Strength (psi) Material AlO3 SiC (sintered) Si3N4...
-
Heal (2001, p. 1) notes that when Frederick Law Olmsted, the developer of New York Citys Central Park, was asked how the city would pay for the park, he responded that its presence would raise...
-
Discuss how equity can be viewed as an option. Who has the option, and what decision can they make?
-
Explain this statement: Using leverage has both good and bad effects.
-
Explain the following terms: incremental cash flow, sunk cost, opportunity cost, externality, cannibalization, and complementary project.
-
What is a syndicated loan? What are its advantages and disadvantages?
-
What cash flows are discounted in the FCFE model, and what is the discount rate?
-
How would you use simple chemical tests to distinguish between the following pairs of compounds? (a) Sodium stearate and p-dodecylbenzenesulfonate (b) Beeswax and "paraffin wax" (c) Trimyristin and...
-
1. What is the semi-annually compounded interest rate if $200 accumulates to $318.77 in eight years? Answer in percentage with two decimal places. 2. What is the quarterly compounded interest rate if...
-
A certain object has a mass of 100 kg and is acted on by a force f(t) = 500[2 e t sin(5t)] N. The mass is at rest at t = 0. Determine the objects velocity at t = 5 s.
-
The equation for the voltage (t) across a capacitor as a function of time is where i(t) is the applied current and Q 0 is the initial charge. Suppose that C = 10 7 F and that Q 0 = 0. Suppose the...
-
A rockets mass decreases as it burns fuel. The equation of motion for a rocket in vertical flight can be obtained from Newton s law, and it is where T is the rockets thrust and its mass as a function...
-
Analyze the Trivago s search engine marketing strategy ( search engine optimization and paid search marketing ).
-
REFERS TO THE DIFFERENCE BETWEEN PLANED AND ACTUAL PERFORMANCE This refers to the difference between the actual performance and the planned performance.
-
List the different phases of a Project and give a brief explanation of each phase. You can find this information www . pmi.org .

Study smarter with the SolutionInn App


