The mechanism for the gas-phase reaction of nitrogen dioxide with carbon monoxide to form nitric oxide and
Question:
The mechanism for the gas-phase reaction of nitrogen dioxide with carbon monoxide to form nitric oxide and carbon dioxide is thought to be
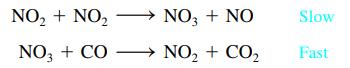
Write the rate law expected for this mechanism. What is the overall balanced equation for the reaction?
Transcribed Image Text:
NO₂ + NO₂ NO3 + CO → NO3 + NO NO₂ + CO₂ Slow Fast
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 90% (10 reviews)

Answered By

Aun Ali
I am an Associate Member of Cost and Management Accountants of Pakistan with vast experience in the field of accounting and finance, including more than 17 years of teaching experience at university level. I have been teaching at both undergraduate and post graduate levels. My area of specialization is cost and management accounting but I have taught various subjects related to accounting and finance.
5.00+
13+ Reviews
32+ Question Solved
Related Book For 

Chemistry
ISBN: 9781305957404
10th Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Question Posted:
Students also viewed these Engineering questions
-
Nitrogen dioxide, NO2, decomposes upon heating to form nitric oxide and oxygen according to the following equation: 2NO2(g) 2NO(g) + O2(g) At the beginning of an experiment, the concentration of...
-
Like nickel, iron reacts with carbon monoxide to form a compound having the formula M(CO)n that obeys the 18-electron rule. What is the value of n in the formula Fe(CO)n?
-
Nitrogen dioxide reacts with carbon monoxide by the overall equation NO2(g)+ CO(g) NO(g) + CO2(g) At a particular temperature, the reaction is second order in NO2 and zero order in CO. The rate...
-
What is the advantage of using computing to simulate an automobile crash test as opposed to actually staging a crash?
-
Explain the many-to-many communication model and why it is important for marketers today.
-
Jones, Jackman and Johnson are partners in the consulting firm of Triple J Partners. The balance sheet of the partnership as at 30 June 2015 is set out below. It was agreed that all profits be...
-
Which of the following procedures is not appropriate for identifying laws and regulations that management may have overlooked? a. Discussing laws and regulations with the entity's chief financial...
-
(Conversion of Bonds) Gabel Company has bonds payable outstanding in the amount of $400,000, and the Premium on Bonds Payable account has a balance of $6,000. Each $1,000 bond is convertible into 20...
-
1. An 88.3-kg refrigerator rests on the floor. How much work is required to move it at constant speed for 2 23 m along the floor against a friction force of 216.1 N? 2. A hammerhead of mass in - 2.35...
-
Your task is write the following functions: 1. create file o takes filename as a parameter. The function creates an empty file. An example is shown below: o create_file("myfile.txt") File myfile....
-
One reason suggested for the instability of long chains of silicon atoms is that the decomposition involves the transition state shown below: The activation energy for such a process is 210 kJ/mol,...
-
A certain substance, initially present at 0.0800 M, decomposes by zero-order kinetics with a rate constant of 2.50 10 -2 mol/L ? s. Calculate the time (in seconds) required for the system to reach a...
-
Calculate the wavelength of a photon needed to excite a transition between neighbouring energy levels of a harmonic oscillator of effective mass equal to that of an oxygen atom (15.9949 u) and force...
-
If Edward purchases $150 worth of casualty insurance at $1.25 per $1,000 of coverage, how much coverage does he have? Explain
-
Renegade Sports have issued 5 year bonds to raise capital for expansion into new markets. The bonds pay coupons annually at an effective annual rate of 10% pa. The face amount of the bonds is...
-
Calculate the operating profit margin ratio given the following financial data: Net Farm Income from Operations $ $110,000 Net Farm Income before Taxes $130,000 Net Farm Income after Taxes $120,000...
-
How Does Lizabeths perspective impact the tone of Marigolds? How do defining moments impact a persons life over time? What is Lizabeths defining moment and how does it impact her as an adult?
-
How is days to collect accounts receivable determined? 365 days divided by accounts receivable turnover. Net sales divided by 365. Net sales divided by average net trade receivables. Accounts...
-
The beam emerging from a ruby laser passes through a circular aperture 5.0 mm in diameter. (a) If the spread of the beam is limited only by diffraction, what is the angular spread of the beam? (b) If...
-
The packaging division of a company having considered several alternative package designs for the company's new product has finally brought down their choices to two designs of which only one has to...
-
Propose a plausible mechanism for each of the following hydrolysis reactions: (a) (b) (c) (d) EtO OEt * * + 2 ELOH (b) N. * .N'
-
Propose a plausible mechanism for the reaction below: [H2SO4] N- - . -N.
-
As shown above, methenamine is hydrolyzed in aqueous acid to produce formaldehyde and ammonia. Draw a mechanism showing formation of one molecule of formaldehyde (the remaining five molecules of...
-
Energy market contains three generation companies and three consumers. The energy bids and offers submitted are presented in the following tables. Plot the supplies and consumer curves. Determine the...
-
During a three - year period, Appliances, Inc., performed electrical heating and plumbing work for Yost Construction worth approximately $ 7 , 0 0 0 . Yost never paid Appliances for any of these...
-
ABC and BCD Joint Corporation is a division of a major corporation. Last year the division had total sales of SAR 8 5 , 7 8 0 , 0 0 0 , net operating income of SAR 8 , 6 9 7 , 5 7 0 , and average...

Study smarter with the SolutionInn App


