The rate of the reaction depends only on the concentration of nitrogen dioxide below 225 C.
Question:
The rate of the reaction
![]()
depends only on the concentration of nitrogen dioxide below 225οC. At a temperature below 225οC, the following data were collected:
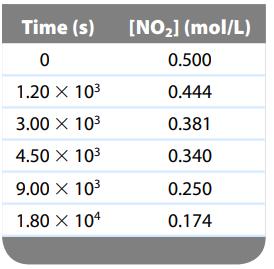
Determine the rate law, the integrated rate law, and the value of the rate constant. Calculate [NO2] at 2.70 × 104 s after the start of the reaction.
Transcribed Image Text:
NO₂(g) + CO(g) NO(g) + CO₂(g)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 40% (5 reviews)
Solution a The rate law is rate k on time t b The integ...View the full answer

Answered By

James Warinda
Hi! I’m James Otieno and I'm an experienced professional online tutor with countless hours of success in tutoring many subjects in different disciplines. Specifically, I have handled general management and general business as a tutor in Chegg, Help in Homework and Trans tutor accounts.
I believe that my experience has made me the perfect tutor for students of all ages, so I'm confident I can help you too with finding the solution to your problems. In addition, my approach is compatible with most educational methods and philosophies which means it will be easy for you to find a way in which we can work on things together. In addition, my long experience in the educational field has allowed me to develop a unique approach that is both productive and enjoyable.
I have tutored in course hero for quite some time and was among the top tutors awarded having high helpful rates and reviews. In addition, I have also been lucky enough to be nominated a finalist for the 2nd annual course hero award and the best tutor of the month in may 2022.
I will make sure that any student of yours will have an amazing time at learning with me, because I really care about helping people achieve their goals so if you don't have any worries or concerns whatsoever you should place your trust on me and let me help you get every single thing that you're looking for and more.
In my experience, I have observed that students tend to reach their potential in academics very easily when they are tutored by someone who is extremely dedicated to their academic career not just as a businessman but as a human being in general.
I have successfully tutored many students from different grades and from all sorts of backgrounds, so I'm confident I can help anyone find the solution to their problems and achieve
0.00
0 Reviews
10+ Question Solved
Related Book For 

Chemistry
ISBN: 9781305957404
10th Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Question Posted:
Students also viewed these Engineering questions
-
The rate of the reaction NO2(g) + CO(g) NO(g) + CO2(g) depends only on the concentration of nitrogen dioxide at temperatures below 225oC. At a temperature below 225oC, the following data were...
-
The rate of the reaction O(g) + NO2(g) NO(g) + O2(g) was studied at a certain temperature. This reaction is one step of the nitric oxide catalyzed destruction of ozone in the upper atmosphere. a. In...
-
The rate of the reaction CH3COOC2H5(aq) + OH (aq) CH3COO(aq) + C2H5OH(aq) Was measured at several temperatures, and the following data were collected: Temperature (oC) k (M-1 s-1)...
-
Write Python code that prompts the user to enter his or her age and assigns the users input to an integer variable named age.
-
What do you believe will be the strongest motivations behind the purchase of an electric car? Do you think the Ontario incentive program is attractive? Do you think this rebate program will cause a...
-
The number of messages sent to a computer bulletin board is a Poisson random variable with a mean of 5 messages per hour. (a) What is the probability that 5 messages are received in 1 hour? (b) What...
-
A model fan with wheel diameter 32 in. is tested at a speed of \(1750 \mathrm{rpm}\). The test fluid is air with density \(0.075 \mathrm{lbm} / \mathrm{ft}^{3}\). At its BEP, the fan produces \(8000...
-
A linear program has a maximum profit of $600. One constraint in this problem is 4X + 2Y 80. Using a computer we find the dual price for this constraint is 3, and there is a lower bound of 75 and an...
-
Using a recent (September 2023- present) Microeconomics related article in the media (the Economist, Globe and Mail, New York Times, etc.), attempt to explain parts or all of it using concepts in...
-
The audit firm of DCG, LLP is performing an audit of Argo Co., a publicly traded company, for the year ending December 31, 2014. You have been given the assignment of assisting in the planning...
-
Hydrogen reacts explosively with oxygen. However, a mixture of H 2 and O 2 can exist indefinitely at room temperature. Explain why H 2 and O 2 do not react under these conditions.
-
The type of rate law for a reaction, either the differential rate law or the integrated rate law, is usually determined by which data is easiest to collect. Explain.
-
Consider the consumptionsaving problem of Section 2.1.2. How would you formulate the problem using multistage stochastic programming with recourse or stochastic dynamic programming?
-
What is the difference between a PHYSICAL test and a PSYCHOMOTOR ABILITIES test? Please also provide an example to with your explanations. (4)
-
Discuss the benefits that your company would receive from nurturing, enabling and empowering its talent
-
What are some ways to improve time management skills for junior leaders
-
Define and describe the pathophysiology and risk factors of Type One Diabetes mellitus.
-
Can you explain the use of Nursing Home Compare and State Operations Manual?
-
About 8.0 106 tons of urea [(NH2)2 CO] are used annually as a fertilizer. The urea is prepared at 200C and under high-pressure conditions from carbon dioxide and ammonia (the products are urea and...
-
Use critical values to test the null hypothesis H0: 1 2 = 20 versus the alternative hypothesis H0: 1 2 20 by setting a equal to .10, .05, .01, and .001. How much evidence is there that the...
-
Draw all constitutionally isomeric ethers with molecular formula C 4 H 10 O. Provide a common name and a systematic name for each isomer.
-
Starting with cyclohexene and using any other reagents of your choice, show how you would prepare each of the following compounds. a. b. c. OH OMe
-
When 1, 4-dioxane is heated in the presence of HI, compound A is obtained: a. Draw the structure of compound A. b. If one mole of dioxane is used, how many moles of compound A are formed? c. Show a...
-
In what ways do organizations integrate sustainability principles and circular economy concepts into their innovation strategies, including cradle-to-cradle product design, resource recovery...
-
On December 31, 2022, Skysong Inc. owns a machine with a carrying amount of $824,000. The original cost and accumulated depreciation for the machine on this date are as follows: Machine $1,400,000...
-
discuss the following terms Concepts of Motivation at work place 2) Six Real life situations of Motivation at work place 3) Benefits observed of Motivation at work place 4) Limitations observed of...

Study smarter with the SolutionInn App


