The structures of adrenaline and aspirin are shown below: Label adrenaline and aspirin as a weak acid
Question:
The structures of adrenaline and aspirin are shown below:
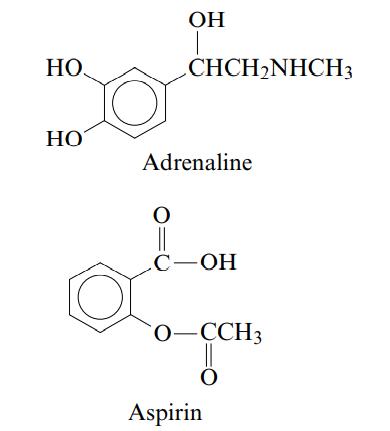
Label adrenaline and aspirin as a weak acid or a weak base. Explain how you can determine this from the structures.
Transcribed Image Text:
НО. HO OH O CHCH₂NHCH3 Adrenaline C-OH O-CCH3 Ö Aspirin
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 70% (10 reviews)
Solution My explanation is that 1 We can tell that adrenaline is ...View the full answer

Answered By

James Warinda
Hi! I’m James Otieno and I'm an experienced professional online tutor with countless hours of success in tutoring many subjects in different disciplines. Specifically, I have handled general management and general business as a tutor in Chegg, Help in Homework and Trans tutor accounts.
I believe that my experience has made me the perfect tutor for students of all ages, so I'm confident I can help you too with finding the solution to your problems. In addition, my approach is compatible with most educational methods and philosophies which means it will be easy for you to find a way in which we can work on things together. In addition, my long experience in the educational field has allowed me to develop a unique approach that is both productive and enjoyable.
I have tutored in course hero for quite some time and was among the top tutors awarded having high helpful rates and reviews. In addition, I have also been lucky enough to be nominated a finalist for the 2nd annual course hero award and the best tutor of the month in may 2022.
I will make sure that any student of yours will have an amazing time at learning with me, because I really care about helping people achieve their goals so if you don't have any worries or concerns whatsoever you should place your trust on me and let me help you get every single thing that you're looking for and more.
In my experience, I have observed that students tend to reach their potential in academics very easily when they are tutored by someone who is extremely dedicated to their academic career not just as a businessman but as a human being in general.
I have successfully tutored many students from different grades and from all sorts of backgrounds, so I'm confident I can help anyone find the solution to their problems and achieve
0.00
0 Reviews
10+ Question Solved
Related Book For 

Chemistry
ISBN: 9781305957404
10th Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Question Posted:
Students also viewed these Engineering questions
-
Explain how you can impose your own limits on credit card spending.
-
Explain how you can determine where f'(x)is increasing or decreasing by examining the graph of y=f{x). In Problem assume that f is a polynomial.
-
Explain how you can use size-quantized quantum dots to create a protein with a barcode that can be read using light.
-
Consider the following recursive function, which is related to a famous unsolved problem in number theory, known as the Collatz problem, or the 3n+1 problem: public static void collatz(int n) {...
-
What are the insureds major duties after a loss?
-
In the 2009 Current Population Survey conducted by the U.S. Census Bureau, two average household incomes were reported: $49,777 and $67,976. One of these averages is the mean and the other is the...
-
A simply supported uniform beam of length \(l\) carries a mass \(M\) at the center of the beam. Assuming \(M\) to be a point mass, obtain the frequency equation of the system.
-
Lauren Barton, a single mother with three children, lived in Portland, Oregon. Cynthia VanHorn also lived in Oregon until she moved to New York City to open and operate an art gallery. VanHorn asked...
-
Write BFS and DFS for a graph: What would be BFS and DFS traversal for the below graphs. Write the nodes for BFS and DFS. Start at node A. A B D C F E G
-
On January 1, 2020, Innovus, Inc., acquired 100 percent of the common stock of ChipTech Company for $670,000 in cash and other fair-value consideration. ChipTechs fair value was allocated among its...
-
Consider a 0.10-M H 2 CO 3 solution and a 0.10-M H 2 SO 4 solution. Without doing any detailed calculations, choose one of the following statements that best describes the [H + ] of each solution and...
-
Of the hydrogen halides, only HF is a weak acid. Give a possible explanation.
-
Do you believe you can do a better job of working or studying than you are currently doing? Do you think you would get more pay and benefits or better grades if you did a better job? Do you care...
-
An endowment fund is set up with a donation of $110,000. If it earns 4.1% compounded monthly, for how long will it sustain end-of- month withdrawals of $1,300? (Include the final smaller withdrawal.)...
-
Defined benefit obligation, 31 December 20X1 Benefits paid to retirees in 20X2 during the year Current service cost for 20X2 Actual return on plan assets for 20X2 Fair value of plan assets, 31...
-
At December 31, Hawke Company reports the following results for its calendar year. Cash sales Credit sales $ 280,000 $700,000 In addition, its unadjusted trial balance includes the following items....
-
a. Suppose the monthly income of an individual increases from Rs 20,000 to Rs 25,000 which increases his demand for clothes from 40 units to 60 units. Calculate the income elasticity of demand. b....
-
There are six types of digital media channels: Search engine marketing, Online PR, Online partnerships, Interactive advertising, Opt-in email marketing, and Social media marketing. Recognize and...
-
(a) What rms current is drawn by a 4200-W electric room heater when running on 120 V rms? (b) What is the power dissipation by the heater if the voltage drops to 105 V rms during a brownout? Assume...
-
Discuss the concept of the looking-glass self. how do you think others perceive you? do you think most people perceive you correctly?
-
In plants, terpenes are biosynthesized by a pathway that involves loss of CO2 from 3-phosphornevalonate 5-diphosphate to yield isopentenyl diphosphate. Use curved arrows to show the mechanism of this...
-
Propose a structure for a compound C 6 H 12 O 2 that dissolves in dilute NaOH and shows the following 1 H NMR spectrum: 1.08 (9 H, singlet), 2.2 (2 H, singlet), and 11.2 (1 H, singlet).
-
What spectroscopic method could you use to distinguish among the following three isomeric acids? Tell what characteristic features you would expect for eachacid. CH3(CH213CO2H (CH/2CHCH-cO2H...
-
The following situation is an example for a fire detection and suppression system in an office building. The Initiating Event (IE) for the Event Tree is "fire starts", with a frequency of once every...
-
A one cubic foot sample of a borrow-source clay weighed 88 lbs. If the specific gravity of solids was measured as 2.70, and the clay was found to be 10 percent saturated, determine the water content...
-
Evaluate the use of novel catalytic systems, such as structured catalysts, catalytic distillation, and multifunctional catalysts, in achieving process intensification, discussing the effects on...

Study smarter with the SolutionInn App


