What type of molecular orbital would result from the in-phase combination of two d xz atomic orbitals
Question:
What type of molecular orbital would result from the in-phase combination of two dxz atomic orbitals shown below? Assume the x-axis is the internuclear axis.
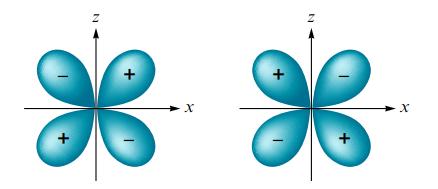
Transcribed Image Text:
z Z X X X
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 70% (10 reviews)
When the two electrons come together and they are out of ...View the full answer

Answered By

Hardik Dudhat
I am semi-qualified Chemical Engineering ,I have scored centum in accounting in my senior secondary and in my graduation. I have always helped my fellow students with their concerns on the subject, i have tutored on various tutoring sites in the past and also have taken home tuitions for degree and MBA students. As a tutor, I don't want my students to just get a solution, I want them to understand the concept and never have a doubt in that area thereon and i believe in excelling and not in educating.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Chemistry
ISBN: 9781305957404
10th Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Question Posted:
Students also viewed these Engineering questions
-
The following sketches show the atomic orbital wave functions (with phases) used to construct some of the MOs of a homo-nuclear diatomic molecule. For each sketch, determine the type of MO that will...
-
The sp2 hybrid atomic orbitals have the following general form: where Ïs, Ïpx, and Ïpy represent orthonormal (normalized and orthogonalized) atomic orbitals. Calculate the values of A...
-
Assume that the cyanide ion, CN, has molecular orbitals similar to those of a homonuclear diatomic molecule. Write the configuration and bond order of CN. Is a substance of the ion diamagnetic or...
-
Roles define the manager. Do you agree or disagree with this statement? Discuss what you think managers do.
-
Why do firms divest assets?
-
Here are comparative statements of financial position for Syal SE. Additional information: 1. Net income for 2020 was 103,000. 2. Depreciation expense was 32,000. 3. Cash dividends of 45,000 were...
-
Late in December 1999, your public accounting firm accepted an audit engagement at Fine Jewelers, Inc., a corporation that maintains a diamond wholesale store in New York City and retail jewelry...
-
Strong Tool Company has been considering purchasing a new lathe to replace a fully depreciated lathe that will last 5 more years. The new lathe is expected to have a 5-year life and depreciation...
-
Sunlight at 0.64 m reflected by the Earth is absorbed by a nadir viewing satellite sensor at an altitude of 800 km. The sensor has a circular detector with a radius of 1 mm and a bandwidth of 0.1 m...
-
Jogger Shoe Company is trying to decide whether to make a change in its most popular brand of running shoes. The new style would cost the same to produce and be priced the same, but it would...
-
Using molecular orbital theory, explain why the removal of an electron from O 2 strengthens bonding, whereas the removal of an electron from N 2 weakens bonding.
-
The transport of O 2 in the blood is carried out by hemoglobin. Carbon monoxide (CO) can interfere with O 2 transport because hemoglobin has a stronger affinity for CO than for O 2 . If CO is...
-
4 + (-7) Find the sum by hand.
-
A 30 year mortgage loan is made at an interest rate of 3.5% APR. The borrower decides to pay $1,000 each month instead of the minimum payment. How many years will it take for the loan to be paid off?...
-
Motherboards may contain which three of the following ports on the I/O panel? a. SATAe b. USB ports c. PCIe ports d. HDMI, DVI, and/or DisplayPort e. RJ-45 LAN port
-
How would a professional option seller utilize the various option greeks to manage their underlying risk exposure?
-
"How does a company's liquidity position impact its ability to meet short-term obligations and maintain financial stability?"
-
Lets compare a savings plan that pays 6% simple interest versus another plan that pays 6% annual interest compounded quarterly. If we deposit $8,000 into each savings account, how much money will we...
-
A 200-W infrared laser emits photons with a wavelength of 2.0 106 m, and a 200-W ultraviolet light emits photons with a wavelength of 7.0 108 m. (a) Which has greater energy, a single infrared...
-
As indicated by mutual fund flows, investors tend to beat the market seek safety invest in last year's winner invest in last years loser
-
What is the difference between a transition state and an intermediate?
-
Draw an energy diagram for a one-step reaction with Keq < 1. Label the parts of the diagram corresponding to reactants, products, transition state, G, and G++. Is G positive or negative?
-
Draw an energy diagram for a two-step reaction with Keq > 1. Label the overall G, transition states, and intermediate. Is G positive or negative?
-
The Step Company has the following Information for the year just ended: Sales in units Budget 15,000 Actual 14,000 Sales $150,000 $147,000 Less: Variable Expenses 90,000 82,600 Contribution Margin $...
-
Question 2 (2 marks) The year level coordinators for Years 7 to 11 at the school are Amy (A), Brian (B), Claire (C), Daisy (D) and Ellie (E). A faulty telephone system means that some of these...
-
On a certain farm alfalfa yields have been 4.3, 7.2, 5.6, and 3.6 tons per acre in the last 4 years. What is the expected value for the farm's alfalfa yield, if each past result is given equal weight...

Study smarter with the SolutionInn App


