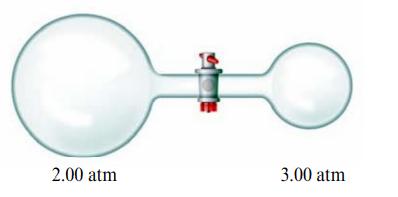
You have helium gas in a two-bulbed container connected by a valve as shown below. Initially the
Question:
You have helium gas in a two-bulbed container connected by a valve as shown below. Initially the valve is closed. When the valve is opened, will the total pressure in the apparatus be less than 5.00 atm, equal to 5.00 atm, or greater than 5.00 atm? Explain your answer.
Transcribed Image Text:
2.00 atm 3.00 atm
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (12 reviews)
It is because of the simple algebraic relation When 500 atm i...View the full answer

Answered By

James Warinda
Hi! I’m James Otieno and I'm an experienced professional online tutor with countless hours of success in tutoring many subjects in different disciplines. Specifically, I have handled general management and general business as a tutor in Chegg, Help in Homework and Trans tutor accounts.
I believe that my experience has made me the perfect tutor for students of all ages, so I'm confident I can help you too with finding the solution to your problems. In addition, my approach is compatible with most educational methods and philosophies which means it will be easy for you to find a way in which we can work on things together. In addition, my long experience in the educational field has allowed me to develop a unique approach that is both productive and enjoyable.
I have tutored in course hero for quite some time and was among the top tutors awarded having high helpful rates and reviews. In addition, I have also been lucky enough to be nominated a finalist for the 2nd annual course hero award and the best tutor of the month in may 2022.
I will make sure that any student of yours will have an amazing time at learning with me, because I really care about helping people achieve their goals so if you don't have any worries or concerns whatsoever you should place your trust on me and let me help you get every single thing that you're looking for and more.
In my experience, I have observed that students tend to reach their potential in academics very easily when they are tutored by someone who is extremely dedicated to their academic career not just as a businessman but as a human being in general.
I have successfully tutored many students from different grades and from all sorts of backgrounds, so I'm confident I can help anyone find the solution to their problems and achieve
0.00
0 Reviews
10+ Question Solved
Related Book For 

Chemistry
ISBN: 9781305957404
10th Edition
Authors: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Question Posted:
Students also viewed these Engineering questions
-
Two rigid tanks are connected by a valve as shown in the accompanying figure. Tank A is insulated and contains 0.1 m3 of steam at 500 kPa and 90% quality. Tank B is uninsulated and contains 2 kg of...
-
Helium gas is compressed from 1 atm and 25C to a pressure of 10 atm adiabatically. The lowest temperature of helium after compression is (a) 25C (b) 63C (c) 250C (d) 384C (e) 476C
-
The gauge pressure in a helium gas cylinder is initially 28atm. After many balloons have been blown up, the gauge pressure has decreased to 5 atm. What fraction of the original gas remains in the...
-
Return to Better Mousetraps in Problem 18. Suppose the firm can cut its requirements for working capital in half by using better inventory control systems. By how much will this increase project NPV?...
-
Describe how an agent can be liable to the principal.
-
Constant pressure lines in the superheated region of the Mollier diagram have (a) a positive sloped (b) a negative slope (c) zero slope (d) both positive and negative slope
-
Superchef World Class Kitchens supplies two varieties of kitchen benchtops, Solid Surface and Laminate. The projected operating data for the month of September are as follows. Required Prepare a...
-
Eastman Publishing Company is considering publishing an electronic textbook on spreadsheet applications for business. The fixed cost of manuscript preparation, textbook design, and Web site...
-
Assume the zero-coupon yields on default-free securities are as summarized in the following table: Maturity 1 year 2 years 3 years 4 years 5 years Zero-Coupon Yields 3.20% 3.60% 3.80% 4.00% 4.20%...
-
Frontier Partners, a management consulting firm, has the following condensed budget for 2017: Frontier has a single direct-cost category (professional labor) and a single indirect-cost pool (client...
-
If you have any two gases in different containers that are the same size at the same pressure and same temperature, what is true about the moles of each gas? Why is this true?
-
If you release a helium balloon, it soars upward and eventually pops. Explain this behavior.
-
What does a financial manager do? How can he or she monitor a firms financial success?
-
You stand at the edge of a cliff that's 200 m high. You have two rocks, one of which you give a kick so it moves horizontally off the edge at a speed of 5 m/s, the other of which you throw at a speed...
-
What is the Knowledge Base? * A . The Knowledge Base contains previously generated prompts that you save in your All outputs. B . The Knowledge Base contains website scans for tone of voice that you...
-
A speculator who has a of wheat to position in wheat futures wants the futures price in the future. a. long; increase b. long; decrease c. short; increase d. long; stay the same e. short; stay the...
-
12. EMF produced in a coil A circular coil of 126 turns and radius 8 cm is placed in and perpendicular to a magnetic field of intensity 0.3 T. If the intensity is reduced to zero in 0.008 s. What EMF...
-
Which of the following permissions are automatically granted to a user? O Any piece that touches the underlying system O Change items in the database O Enabling extensions O All permissions need to...
-
A cross was made between two pea plants, TtAa and Ttaa, where T = tall, t - dwarf, A = axial, and a = terminal. What is the probability that the first three offspring will be tall with axial flowers...
-
What are some of the features of the Unified Process (UP)?
-
Predict the geometry at the carbon of these compounds: a) H-C=N: b) 0: H-C-0-H c) H T CI-C-H T CI
-
Predict the geometry of the following compounds at the indicated atoms: a) H H-C-0-H H At the carbon At the oxygen b) H. H C-N-H At the carbon At the nitrogen c) H. C=O-H H At the carbon At the oxygen
-
Predict the direction of the dipole moments of these compounds: a) H H b) F H-C-H H H H-C-C=N H
-
Using Fourier Transforms, solve the following integral Equation for ((t) f (t) - ( ( ( t -u) ue - 2 u H (u) du = foo_flt -4t e H(t)
-
Q1: what is the net operating income under the variable and absorption cost system. A: ACER company Product sales: 1,000 units at $10 each Variable manufacturing costs: $5.50 per unit Fixed...
-
Solve for 2 and graph the solution on the number line below. 245x+9 or 5x +9 69

Study smarter with the SolutionInn App


