Determine the total apparent specific heat at constant pressure (c p,mix in kJ/kg K) for a
Question:
Determine the total apparent specific heat at constant pressure (cp,mix in kJ/kg · K) for a fuel–air reactant mixture containing 1 kmol CH4, 2.5 kmol O2, and 9.4 kmol N2 at 500 K and 1 atm. Use specific heat values for each species from Table E.1.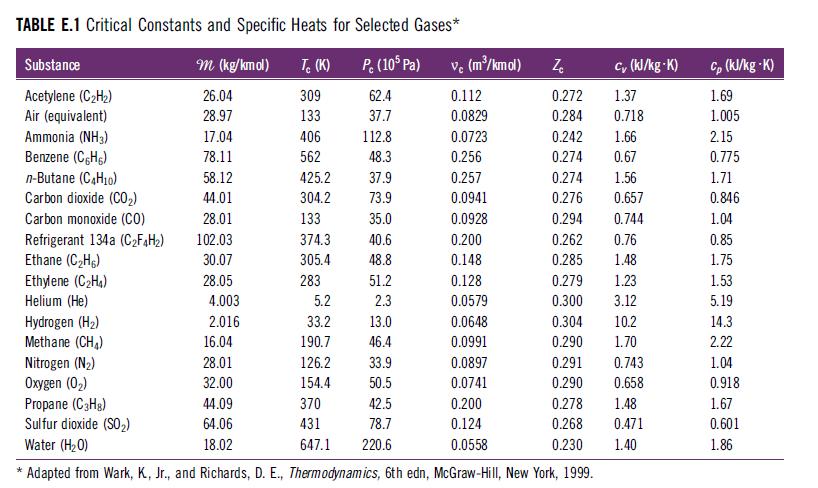
Transcribed Image Text:
TABLE E.1 Critical Constants and Specific Heats for Selected Gases* P. (105 Pa) 62.4 37.7 Substance Acetylene (C₂H₂) Air (equivalent) Ammonia (NH3) Benzene (CH) n-Butane (C4H10) Carbon dioxide (CO₂) Carbon monoxide (CO) * Refrigerant 134a (C₂F4H₂) Ethane (C₂Hg) Ethylene (C₂H4) Helium (He) m (kg/kmol) 26.04 28.97 17.04 78.11 58.12 44.01 28.01 102.03 30.07 28.05 4.003 2.016 Tc (K) 309 133 406 562 425.2 304.2 133 374.3 305.4 283 5.2 33.2 190.7 126.2 154.4 370 431 647.1 112.8 48.3 37.9 73.9 35.0 40.6 48.8 51.2 2.3 13.0 46.4 33.9 50.5 42.5 78.7 220.6 vc (m³/kmol) 0.112 0.0829 0.0723 0.256 0.257 0.0941 0.0928 0.200 0.148 0.128 0.0579 cy (kJ/kg-K) 1.37 0.718 0.242 1.66 0.274 0.67 0.274 1.56 0.276 0.657 0.294 0.744 0.262 0.76 0.285 1.48 Zc 0.272 0.284 0.279 1.23 0.300 3.12 Hydrogen (H₂) 0.0648 Methane (CH₂) 16.04 0.0991 Nitrogen (N₂) 28.01 0.0897 Oxygen (0₂) 32.00 0.0741 Propane (C3H8) 44.09 0.200 Sulfur dioxide (SO₂) 64.06 0.124 Water (H₂O) 18.02 0.0558 Adapted from Wark, K, Jr., and Richards, D. E., Thermodynamics, 6th edn, McGraw-Hill, New York, 1999. 0.304 0.290 0.291 0.290 10.2 1.70 0.743 0.658 0.278 1.48 0.268 0.471 0.230 1.40 Cp (kJ/kg-K) 1.69 1.005 2.15 0.775 1.71 0.846 1.04 0.85 1.75 1.53 5.19 14.3 2.22 1.04 0.918 1.67 0.601 1.86
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (5 reviews)
To determine the total apparent specific heat at constant pressure for the given fuelair mixture we ...View the full answer

Answered By

Joshua Marie Geuvara
I am an academic writer with over 5 years of experience. I write term papers, essays, dissertations, reports, and any other academic paper. My main objective is to produce a high-quality paper free from plagiarism and ensure a student scores an A+. Being a fluent English speaker, I have great communication skills that also enable me to produce excellent papers.
I am conversant with most academic referencing styles (APA, MLA, and Harvard).
You can trust me with your paper and expect nothing less than quality and excellent results. I look forward to meeting with you and, more importantly, developing something that will both make us happy and satisfied.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Thermodynamics Concepts And Applications
ISBN: 9781107179714
2nd Edition
Authors: Stephen R. Turns, Laura L. Pauley
Question Posted:
Students also viewed these Engineering questions
-
Determine the total apparent specific heat at constant volume (c v,mix in kJ/kgK) for a fuelair reactant mixture containing 1 kmol CH 4 , 2.5 kmol O 2 , and 9.4 kmol N 2 at 500 K and 1 atm. What heat...
-
A mixture of ideal gases contains 0.5 kmol of CO 2 , 2 kmol of O 2 , and 7 kmol of N 2 at 700 K. Determine the following quantities: A. The mole fraction of each constituent in the mixture. B. The...
-
Complete the following questions. Submit journal entries in an Excel Template, linked at the bottom of this page, and any written segments in Excels comments function. Do not submit two separate...
-
Format only cells with values greater than maxim 4 tab and select Red color from the pallet (bottom ro Format only cells with values less than minimum and select Yellow color from the pallet (bottom...
-
Obtain IRS Revenue Ruling 59 60 through the IRS web site. Read this Ruling and prepare a memo to your professor explaining how each of its eight valuation factors fits into the theory of business...
-
Gold sells at $1,209.10 per ounce. It has a convenience yield denoted by y and a storage cost u = 4% (per annum continuously compounded). The risk-free interest rate is 3% per annum continuously...
-
Recycling concrete aggregate is an important component of green engineering. The strength of any potential material, expressed in terms of its resilient modulus, must meet standards before it is...
-
Here are data on two companies. The T-bill rate is 4% and the market risk premium is 6%. What would be the fair return for each company, according to the capital asset pricing model(CAPM)? Company...
-
Suppose the economy has the following consumption pattern Sandwich Hotdog Cell phone Super car 2019 Price $7 $3 $200 $5,000 Quantity 500 400 100 3 2020 Price $10 $5 $250 $7,000 Quantity 200 400 100 2...
-
Brothers Mike and Tim Hargenrater began operations of their tool and die shop (H & H Tool, Inc.) on January 1, 2019. The annual reporting period ends December 31. The trial balance on January 1,...
-
A 1-m 3 tank contains nitrogen at 30C and 500 kPa. In an isothermal process, CO 2 is forced into this tank until the pressure is 1000 kPa. What is the mass (kg) of each gas present at the end of this...
-
A 17.3-liter tank contains a mixture of argon, helium, and nitrogen at 298 K. The argon and helium mole fractions are 0.12 and 0.35, respectively. If the partial pressure of the nitrogen is 0.8 atm,...
-
Jeanette bought a small office building with a 25 percent cash payment and a 75 percent mortgage. The total price was $400 000, and her monthly mortgage payment was $3000. The location of the...
-
In Problems 15-18, use Hamilton's plan to apportion the new seats to the existing states. Then increase the number of seats by one and decide whether the Ala bama paradox occurs. Assume that the...
-
Chemistry is taught at five high schools in the Santa Rosa Unified School District. The district has just received a grant of 100 microscopes which are to be apportioned to the five high schools...
-
Use this information to answer the questions in Problems 12-17. How many votes would be necessary for a majority? In voting among three candidates, the outcomes are reported as: (BAC) (CAB) (CBA) 3 2...
-
For the given year, find the standard quotas for the New York City boroughs given in Table 17.5 in Problems 23-28. Assume there are eight council seats. Table 17. 5 1900 Year Total 1790 49 1800 81...
-
Give one example in which you have participated in voting where the count was tabulated by using the tournament method. Your example can be made up or factual, but you should be specific.
-
Describe the differences between descriptive, predictive, and prescriptive analytics.
-
Why should you not model a decision variable as a random variable with a probability distribution?
-
Determine the value of the stoichiometric coefficients for the combustion of coal in oxygen given by the stoichiometric equation: CHN 0:01 O 0:1 S 0:05 + aO 2 = bCO 2 + cH 2 O + dN 2 + eSO 2
-
Determine the value of the stoichiometric coefficients for the combustion of natural gas in air: CH 4 + a(O 2 + 3:76N 2 ) = bCO 2 + cH 2 O + dN 2
-
Using the stoichiometric coefficients you found in exercise 9, determine the molar air-to-fuel ratio (A/F) molar for the combustion of natural gas in air. (A: (A/F) molar = 9.52 kmol of air/kmol of...
-
Hall & Company issues $ 1 0 0 , 0 0 0 , 0 0 0 in 6 % bonds with detachable warrants of 1 0 warrants for each $ 1 , 0 0 0 bond. The bonds are issued at 9 9 . Record the issuance under the following...
-
Assume that hein acquired Dolan Development last year. Hein recorded the following intangible assets on the date of acquisition: Goodwill: $ 1 , 5 0 0 , 0 0 0 Dolan Development trademark: $ 6 0 0 , 0...
-
What is the total amount of interest that will be paid of the loan is carried to term with no extra payments made building cost 3 3 0 0 0 0 . loan started im Dec 2 0 2 3 worh a 7 . 5 % interest rate...

Study smarter with the SolutionInn App


