Determine the total apparent specific heat at constant volume (c v,mix in kJ/kgK) for a fuelair reactant
Question:
Determine the total apparent specific heat at constant volume (cv,mix in kJ/kg·K) for a fuel–air reactant mixture containing 1 kmol CH4, 2.5 kmol O2, and 9.4 kmol N2 at 500 K and 1 atm. What heat transfer is required to cool the mixture from 500 K to 300 K? Use specific heat values for each species from Table E.1.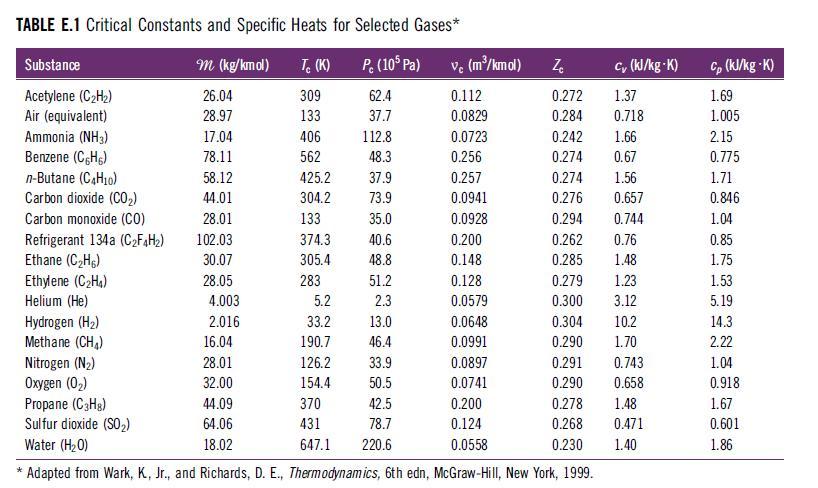
Transcribed Image Text:
TABLE E.1 Critical Constants and Specific Heats for Selected Gases* P. (105 Pa) 62.4 37.7 Substance Acetylene (C₂H₂) Air (equivalent) Ammonia (NH3) Benzene (CH) n-Butane (C4H10) Carbon dioxide (CO₂) Carbon monoxide (CO) Refrigerant 134a (C₂F4H₂) Ethane (C₂H) Ethylene (C₂H₁) Helium (He) m (kg/kmol) 26.04 28.97 17.04 78.11 58.12 44.01 28.01 102.03 30.07 28.05 4.003 2.016 Tc (K) 309 133 406 562 16.04 28.01 32.00 425.2 304.2 133 374.3 305.4 283 5.2 33.2 190.7 126.2 154.4 370 431 647.1 112.8 48.3 37.9 73.9 35.0 40.6 48.8 51.2 2.3 13.0 46.4 33.9 50.5 42.5 78.7 220.6 vc (m³/kmol) 0.112 0.0829 0.0723 0.256 0.257 0.0941 0.0928 0.200 0.148 0.128 0.0579 Ze cy (kJ/kg-K) 0.272 1.37 0.284 0.718 0.242 1.66 0.274 0.67 1.56 0.657 0.274 0.276 0.294 0.262 0.285 Hydrogen (H₂) 0.0648 Methane (CH₂) 0.0991 Nitrogen (N₂) 0.0897 Oxygen (0₂) 0.0741 Propane (C₂H₂) 44.09 0.200 Sulfur dioxide (SO₂) 64.06 0.124 Water (H₂O) 18.02 0.0558 * Adapted from Wark, K, Jr., and Richards, D. E., Thermodynamics, 6th edn, McGraw-Hill, New York, 1999. 0.279 1.23 0.300 3.12 0.744 0.76 1.48 0.304 0.290 10.2 1.70 0.291 0.743 0.290 0.658 0.278 1.48 0.268 0.471 0.230 1.40 Cp (kJ/kg-K) 1.69 1.005 2.15 0.775 1.71 0.846 1.04 0.85 1.75 1.53 5.19 14.3 2.22 1.04 0.918 1.67 0.601 1.86
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 58% (12 reviews)
To determine the total apparent specific heat at constant volume for the given reactant mixture we need to first calculate the apparent specific heat ...View the full answer

Answered By

Deepak Sharma
0.00
0 Reviews
10+ Question Solved
Related Book For 

Thermodynamics Concepts And Applications
ISBN: 9781107179714
2nd Edition
Authors: Stephen R. Turns, Laura L. Pauley
Question Posted:
Students also viewed these Engineering questions
-
Determine the total apparent specific heat at constant pressure (c p,mix in kJ/kg K) for a fuelair reactant mixture containing 1 kmol CH 4 , 2.5 kmol O 2 , and 9.4 kmol N 2 at 500 K and 1 atm. Use...
-
A mixture of ideal gases contains 0.5 kmol of CO 2 , 2 kmol of O 2 , and 7 kmol of N 2 at 700 K. Determine the following quantities: A. The mole fraction of each constituent in the mixture. B. The...
-
A mixture at 500 kPa and 1000 K contains 2 kmol O 2 , 8 kmol N 2 , and 0.2 kmol of H 2 O. Find the apparent molecular weight of the mixture. Determine the apparent specific heat at constant volume...
-
840N with Two cables AB and AC are acting on the pole with forces FAB = 420N and FAC parameters defining the attachment points shown in the table. We want to write the vector FAB in cartesian vector...
-
1. It is important for a forensic accountant to understand the client firms industry as well as local and national economic conditions. a. True b. False 2. A forensic accountant need not consider the...
-
Luzadis Company makes furniture using the latest automated technology. The company uses a job-order costing system and applies manufacturing overhead cost to products based on machine-hours. The...
-
List two ways in which monitoring by the Joint Monitoring Unit and the ACCA monitoring unit differ and give reasons to explain these differences.
-
During 2011, Arthur Corporation reported a net income of $3,059,000. On January 1, Arthur had 2,800,000 shares of common stock outstanding. the company issued an additional 1,680,000 shares of common...
-
Zachary Corporation paid one of its sales representatives $8,500 during the month of March. The rep is paid a base salary plus $14 per unit of product sold. During March, the rep sold 170 units....
-
Find f'(x) for f(x) = 2x + 3x - 1. Be sure to use f'(x) = lim BH to get credit. or f'(x) = lim A-0 f(x+h)-f(x) h f(b)-f(x) b-I
-
A mixture of products of combustion contains the following constituents at 2000 K: 3 kmol of CO 2 , 4 kmol of H 2 O, and 18.8 kmol of N 2 . Determine the following quantities: A. The mole fraction of...
-
Explain in words how the mass-specific and molar-specific enthalpies of an ideal-gas mixture relate to the corresponding properties of the constituent species.
-
A drug manufacturer claims that less than 10% of patients who take its new drug for treating Alzheimers disease will experience nausea. To test this claim, researchers conduct an experiment. They...
-
Use Jefferson's plan in Problems 11-14. Show that it violates the quota rule. State: A Population: 17,179 Number of seats: 132 B 7,500 49,400 D 5,824
-
Use Jefferson's plan in Problems 11-14. Show that it violates the quota rule. State: A Population: 68,500 Number of seats: 100 B 34,700 C 14,800 D 9,500
-
Give one example in which you have participated in voting where the count was tabulated by the Hare voting method. Your example can be made up or factual, but you should be specific.
-
How many ounces of a base metal (no silver) must be alloyed with 100 ounces of \(21 \%\) silver alloy to obtain an alloy that is \(15 \%\) silver?
-
Suppose the number (in millions) of bacteria present in a culture at time \(t\) is given by the formula \[N(t)=2 t^{2}-200 t+1,000\] Use this formula in Problems 57-60. Derive a formula for the...
-
Define the eight characteristics of quality data.
-
CdF2 (s) Cd+ (aq) + 2 F- (aq) 1. A saturated solution of CdF2 is prepared. The equilibrium in the solution is represented above. In the solution [Cd+] eq = 0.0585 M and [F-] eq = 0.117 M. a....
-
It is 2021 and you are a process engineer at a large oil refining company. The world is rapidly moving toward a hydrogen energy economy and your company has been trying to develop an efficient and...
-
What would be the Otto cycle efficiency in Example 6.1 if the engines compression ratio was 10.0? Example 6.1 A new four-cylinder Otto cycle engine has a total displacement of 5000. cm 3 and a...
-
What would be the ideal power produced in Example 6.1 if the engines total displacement was 8000. cm 3 and it was running at 3000. RPM? Example 6.1 A new four-cylinder Otto cycle engine has a total...
-
US exports to China have been rapidly increasing but not fast enough to offset the imports from China. This means that the United States has a/an? Give answer and explain.
-
5) Write Matlab expressions to extract only the elements at odd positions in a vector (i.e. the 1st, 3rd, 5th, 7th..., elements of the vector), regardless of the length of the vector. Make sure your...
-
1. Convert NFA to DFA 91 1 0 a. b. (92) 1 (93) 1. 90 1 0,1 0,1 (92)

Study smarter with the SolutionInn App


