2000 kg of oxygen is in a 1.8 m 3 container maintained at 90 K. Calculate the...
Question:
2000 kg of oxygen is in a 1.8 m3 container maintained at 90 K. Calculate the liquid and vapor masses (kg) and the liquid and vapor volumes (m3). Use the tables in Appendix A.8 for data.
Data From Appendix A.8
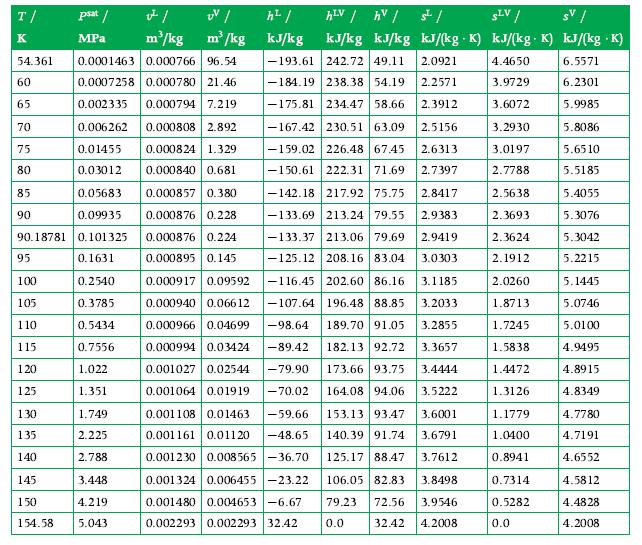
Transcribed Image Text:
T/ K 85 90 54.361 0.0001463 0.000766 96.54 60 0.0007258 0.000780 21.46 65 0.002335 0.000794 7.219 70 0.006262 0.000808 2.892 75 0.01455 0.000824 1.329 80 0.03012 0.000840 0.681 0.05683 0.000857 0.380 0.09935 0.000876 0.228 90.18781 0.101325 0.000876 0.224 0.1631 0.000895 0.145 0.2540 0.3785 0.5434 0.7556 1.022 1.351 95 100 105 110 115 120 125 psat / MPa 130 135 140 145 150 1.749 2.225 2.788 3.448 4.219 154.58 5.043 v²/ UV/ h²/ m³/kg m³/kg kJ/kg hLV / hv / SLV / sv / kJ/kg kJ/kg kJ/(kg K) kJ/(kg K) kJ/(kg .K) 4.4650 6.5571 3.9729 6.2301 3.6072 5.9985 3.2930 5.8086 3.0197 5.6510 2.7788 5.5185 5.4055 5.3076 5.3042 5.2215 5.1445 5.0746 5.0100 4.9495 4.8915 4.8349 4.7780 4.7191 4.6552 4.5812 4.4828 4.2008 0.000917 0.09592 0.000940 0.06612 0.000966 0.04699 0.000994 0.03424 -89.42 -79.90 0.001027 0.02544 0.001064 0.01919 -70.02 0.001108 0.01463 -59.66 0.001161 0.01120 -48.65 0.001230 0.008565-36.70 0.001324 0.006455-23.22 0.001480 0.004653-6.67 0.002293 0.002293 32.42 ملی -193.61 242.72 49.11 2.0921 -184.19 238.38 54.19 2.2571 -175.81 234.47 58.66 2.3912 -167.42 230.51 63.09 2.5156 226.48 67.45 2.6313 -159.02 -150.61 222.31 71.69 2.7397 -142.18 217.92 75.75 2.8417 -133.69 213.24 79.55 2.9383 -133.37 213.06 79.69 2.9419 -125.12 208.16 83.04 3.0303 -116.45 202.60 86.16 3.1185 -107.64 196.48 88.85 3.2033 -98.64 189.70 91.05 3.2855 182.13 92.72 3.3657 173.66 93.75 3.4444 164.08 94.06 3.5222 153.13 93.47 3.6001 140.39 91.74 3.6791 125.17 88.47 3.7612 106.05 82.83 3.8498 79.23 72.56 3.9546 32.42 0.0 4.2008 2.5638 2.3693 2.3624 2.1912 2.0260 1.8713 1.7245 1.5838 1.4472 1.3126 1.1779 1.0400 0.8941 0.7314 0.5282 0.0
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (6 reviews)
From the table for saturated state in Appendix A8 we have The mass fraction of vap...View the full answer

Answered By

Zablon Gicharu
I am an educator who possesses the requisite skills and knowledge due to interacting with students for an extended period. I provide solutions to various problems in step-by-step explanations, a well-thought approach and an understandable breakdown. My goal is to impart more straightforward methodologies and understanding to students for more remarkable achievements.
5.00+
4+ Reviews
10+ Question Solved
Related Book For 

Thermodynamics Fundamentals And Engineering Applications
ISBN: 9780521862738
1st Edition
Authors: William C. Reynolds, Piero Colonna
Question Posted:
Students also viewed these Engineering questions
-
A small pharmaceutical firm plans to manufacture a new drug and has hired you as a consultant to design a condenser to remove the drug from a gasvapor mixture. The mixture, which contains 20 mole% of...
-
A small pharmaceutical firm plans to manufacture a new drug and has hired you as a consultant to design a condenser to remove the drug from a gasvapor mixture. The mixture, which contains 20 mole% of...
-
A small pharmaceutical firm plans to manufacture a new drug and has hired you as a consultant to design a condenser to remove the drug from a gasvapor mixture. The mixture, which contains 20 mole% of...
-
(b) (i) What is the smallest natural number n, such that n!> 2" (ii) Prove that n !> 2" is true any natural number greater than or equal that number. (c) Given a finite set of natural numbers ACN....
-
Whole Life Clinic is a VHWO that has three main programs: Drug rehabilitation Alcohol recovery Weight control Unrestricted public support received during the period was $35,000; revenues from...
-
Vic Corporation operates entirely in the United States but in different industries. It segments the business based on industry. Total sales of the segments, including intersegment sales, are as...
-
The three-parameter corresponding state is concerned with (a) Temperature, pressure and volume (b) Reduced temperature, reduced pressure and reduced volume (c) Critical temperature, critical pressure...
-
Consider the Tragedy of the Commons game from the chapter with two shepherds, A and B, where sA and sB denote the number of sheep each grazes on the common pasture. Assume that the benefit per sheep...
-
fa branch/jump instruction is completed changing the value of PC. (b) What LEGv8 instruction does each of the following 32-bit binary numbers represent? reduces 1 1010011010000000011100110101101 Ease...
-
1. Liam Richardson is the business manager for the Smith & Lyngate Insurance agencies in the state of Maryland. Liam is interested in increasing the number of agents in Baltimore and plans to buy...
-
Consider the system in the figure below. Initially the gas in the cylinder is at high pressure P i and the piston held in place at volume V i with a lock. At time zero the lock is released and the...
-
Imagine turning the system of Exercise 2.13 vertically. Repeat the analysis, which now will involve the height of the piston. Using the same values as in Exercise 2.13(b), with D = 8 cm, plot the...
-
You are asked to referee a grant application, which proposes to determine whether the magnetization of iron is due to "Ampere" dipoles (current loops) or "Gilbert" dipoles (separated magnetic...
-
On November 20, 2016, Rainer Grtner, chief executive officer (CEO) of Daimler Trucks and Buses China Ltd. (DTBC), the Chinese heavy-duty segment of multinational automobile manufacturer Daimler AG...
-
On 1 July 2019, Denman Ltd issued a prospectus offering 160 000 of its ordinary shares, payable $1 on application, $1 on allotment and $2 to be called as and when required. When applications closed...
-
Maitland Ltd has issued 2 000 000 ordinary shares for $4 and 200 000 8% preference shares for $4, all shares being fully paid. On 30 September 2019, at the annual general meeting of the company, a...
-
Bega Ltd was registered as a new company on 2 January 2019. On that day a prospectus was issued inviting applications for 300 000 ordinary shares at $10, payable $2.50 on application, $2.50 on...
-
During the year ended 30 June 2019, the directors of Cooma Ltd paid a final dividend out of retained earnings of $60 000, which had been recommended at the end of the previous financial year. They...
-
Consider the following two events for an individual: A = owns a cell phone B = owns a laptop computer Translate each event into words. (a) Ac (b) A and B (c) A or B (d) A | B (e) B | A
-
U.S. households have become smaller over the years. The following table from the 2010 GSS contains information on the number of people currently aged 18 years or older living in a respondent's...
-
A portable music player is sitting in a docking station (Figure P4.31). The docking station has a mass of 500 g and the player, 100 g. Determine the reaction forces at the two supports. Figure P4.31...
-
Two pots of food are being cooked on a solar cooker (Figure P4.32). The smaller pot weighs 4 lb, and the larger pot weighs 9 lb. Also, due to the thermal expansion of the parabolic reflector, a...
-
Find an example of a structure or machine that has several forces acting on it. (a) Make a clear, labeled drawing of it. (b) Estimate the dimensions and the magnitudes and directions of the forces...
-
Think of a process--it could be as simple as making a peanut butter and jelly sandwich, or changing a light bulb. Using Gliffy, draw a flowchart for your process. What were some of your likes and...
-
Gitano Products operates a job - order costing system and applies overhead cost to jobs on the basis of direct materials used in production ( not on the basis of raw materials purchased ) . Its...
-
How is the cost for a plant asset measured in a cash transaction? In a noncash transaction? Please explain.

Study smarter with the SolutionInn App


