An ideal gas is characterised by the relation pV = NRT where p is the pressure of
Question:
An ideal gas is characterised by the relation pV = NRT where p is the pressure of the gas, V is the volume, T is the temperature, N is the number of moles of gas and R is a constant.a) Calculate the differential dp (T, V)
b) Calculate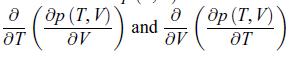
Transcribed Image Text:
8 ( 8p ( T, V ) ) OT 8Ꮴ and 8 ( Ꭷp ( T, V ) ᎥᏙ ᎧᎢ
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 85% (7 reviews)
a To calculate the differential dp T V we first need to find the partial derivatives of p with respe...View the full answer

Answered By

Anik Kumar Bosu
I am expert in Maths, Physics, Chemistry and Biology. I also teach student from 3 years. Students becomes satisfied by my teaching. So, do not worry.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Principles Of Thermodynamics
ISBN: 9781108426091
1st Edition
Authors: Jean-Philippe Ansermet, Sylvain D. Brechet
Question Posted:
Students also viewed these Engineering questions
-
An ideal gas is characterised by the relation pV = NR T as in 1.2 where the pressure p (T, V) is a function of T and V, the temperature T (p, V) is a function of p and V and the volume (T, p) is a...
-
A gas is characterised by the enthalpy H(S, p) = C p T, where C p is a constant (called heat capacity and defined in 5.2), and by pV = NR T, where p is its pressure, V its volume, T its temperature...
-
Thermal Expansion of an Ideal Gas (a) The pressure p, volume V, number of moles n, and Kelvin temperature T of an ideal gas are related by the equation p V = nRT, where R is a constant. Prove that...
-
What are the "two main lines" of distortion of the view of Marx and what does Lenin think the proper view?
-
Record the following annuity and life income activities of Private University: 1. On July 1, 2010, R. W. Fields, emeritus professor of accounting, moved out of the state. Fields donated to the...
-
I bought the old Target store in Barrie on June 12th, 2016. The building had been sitting vacant since Target closed down in 2015. I started using a space in the building as my business office...
-
P(170 < x < 195) The random variable x is normally distributed with mean = 174 and standard deviation = 20. Find the indicated probability.
-
On July 1, 2011, Flashlight Corporation sold equipment it had recently purchased to an unaffiliated company for $480,000. The equipment had a book value on Flashlights books of $390,000 and a...
-
Subprime mortgages and derivatives, bailout of FNMA, Freddie Mac, and AIG, Analysis the following corrective action taken by the Federal Reserve as a result of the crisis: Quantitative easing...
-
For the base case in this section, as a percentage of sales, COGS = 70 percent, SGA = 14 percent, R&D = 2.8 percent. Depreciation, Interest expense are fixed as stated. Tax Rate is 21 percent. 1.a...
-
A basin contains Ns (t) moles of salt dissolved in Nw (t) moles of water. The basin receives fresh water at a constant rate in w. This water is assumed to be thoroughly mixed in the basin so that the...
-
Two of the Massieu functions are functions of the following state variables: 1. 2. The Massieu functions are obtained by performing Legendre transformations of the state function entropy S (U, V)...
-
Use the sales array to answer. The array was declared using the int sales[5] = {10000, 12000, 900, 500, 20000}; statement. The sales[3] += 10; statement will replace the number _____________________....
-
1. An unpolarized light beam is incident on the polarizes of a polarization experiment and the intensity of light beam emerging from the analyzer is measured as 100 Lumens. Now, if the analyzer is...
-
Cheryl is going to purchase a travel trailer priced at $10,500. She has saved $1,000 for a down payment and plans to repay the balance with equal monthly payments. The maximum amount she can afford...
-
A shaft made of mild steel is keyed to a cone clutch assembly. If it has a 2.5 IN diameter and rotates at 1375 RPM. What will be the contact surface size in (MM) if the shaft can take up 5 times its...
-
What should an employee do if they believe they have been subjected to workplace harassment?
-
What is the price of a bond with a coupon rate of 5%, payable semi-annually, a face value of $1000, 15 years to maturity, and a yield to maturity of 4.7%? Enter your response below. Enter your answer...
-
For an analysis of the salaries of your company, you plot the salaries of all employees against the number of years they have worked for the company. You find that plotting the base-10 logarithm of...
-
Ex. (17): the vector field F = x i-zj + yz k is defined over the volume of the cuboid given by 0x a,0 y b, 0zc, enclosing the surface S. Evaluate the surface integral ff, F. ds?
-
A solution of NaOH was standardized by titration of a known quantity of the primary standard, potassium hydrogen phthalate The NaOH was then used to find the concentration of an unknown solution of H...
-
Write the names and abbreviations for each of the prefixes from 10 -24 to 10 24 . Which abbreviations are capitalized?
-
What is the formal concentration (expressed as mol/L = M) of NaCl when 32.0 g are dissolved in water and diluted to 0.500 L?
-
Tonth of June. a. J. Aracel, the owner, invested $245,000 cash, office equipment with a value of $6,700, and $62,000 of drafting equipment to launch the company in exchange for common stock. b. The...
-
On September 1, 2024, the company borrowed $20,000 from a local bank and signed a note. Principal and interest at 12% will be paid on August 31, 2025. Prepare the adjusting entry at December 31.
-
Mia Company uses activity-based costing and reports the following for this year. Activity Cutting Assembly Total Budgeted Cost $ 32,200 86,000 $ 118,200 Activity Cost Driver Machine hours (MH) Direct...

Study smarter with the SolutionInn App


