Calculate the density of gaseous CO 2 at each of the following states:a. 10 bar, 290 K,b.
Question:
Calculate the density of gaseous CO2 at each of the following states:a. 10 bar, 290 K,b. 30 bar, 333 K,c. 100 bar, 410 K,Using the perfect gas model. Compare the values obtained with the values listed in the tables in Appendix A.9. What can you infer?
Data From Appendix A.9
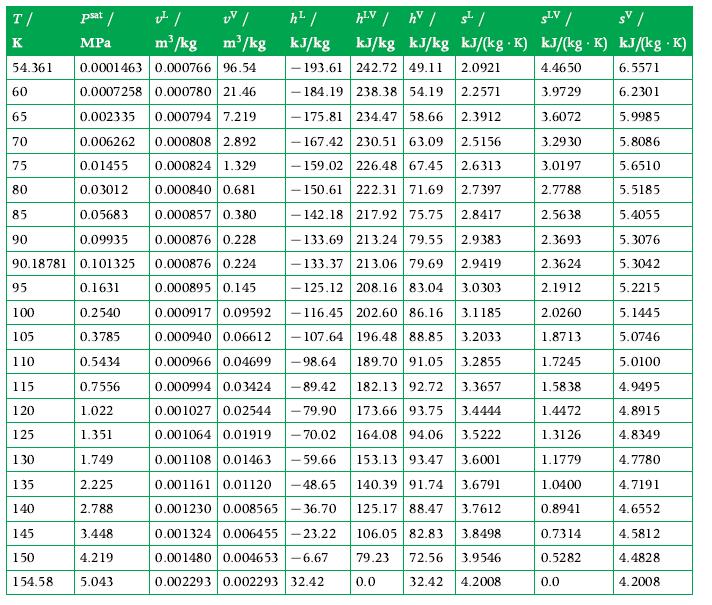
Transcribed Image Text:
T/ K UV/ m³/kg m³/kg 54.361 0.0001463 0.000766 96.54 0.0007258 0.000780 21.46 0.002335 0.000794 7.219 0.000808 2.892 0.000824 1.329 0.006262 0.01455 0.03012 0.000840 0.681 0.05683 0.000857 0.380 0.09935 0.000876 0.228 90.18781 0.101325 0.000876 0.224 0.1631 0.000895 0.145 60 65 70 75 80 85 psat / MPa 90 95 100 0.2540 105 0.3785 110 0.5434 115 0.7556 120 1.022 125 1.351 130 1.749 135 2.225 140 2.788 145 3.448 150 4.219 154.58 5.043 h² / kJ/kg hLV / hv / 5¹/ SLV / SV/ kJ/kg kJ/kg kJ/(kg K) kJ/(kg. K) kJ/(kg .K) 4.4650 6.5571 3.9729 6.2301 3.6072 5.9985 3.2930 5.8086 3.0197 5.6510 2.7788 5.5185 2.5638 5.4055 2.3693 5.3076 2.3624 5.3042 2.1912 5.2215 2.0260 5.1445 1.8713 5.0746 1.7245 5.0100 1.5838 4.9495 1.4472 4.8915 1.3126 4.8349 1.1779 4.7780 1.0400 4.7191 0.8941 4.6552 0.7314 4.5812 0.5282 4.4828 4.2008 193.61 242.72 49.11 2.0921 184.19 238.38 54.19 2.2571 175.81 234.47 58.66 2.3912 - 167.42 230.51 63.09 2.5156 -159.02 226.48 67.45 2.6313 -150.61 222.31 71.69 2.7397 -142.18 217.92 75.75 2.8417 2.9383 -133.69 213.24 79.55 133.37 213.06 79.69 2.9419 -125.12 208.16 83.04 3.0303 0.000917 0.09592 -116.45 202.60 86.16 3.1185 0.000940 0.06612 -107.64 196.48 88.85 3.2033 182.13 92.72 3.3657 0.000966 0.04699 -98.64 189.70 91.05 3.2855 0.000994 0.03424 -89.42 0.001027 0.02544 - 79.90 0.001064 0.01919 - 70.02 173.66 93.75 3.4444 164.08 94.06 3.5222 153.13 93.47 3.6001 0.001108 0.01463 -59.66 0.001161 0.01120 -48.65 3.6791 140.39 91.74 0.001230 0.008565-36.70 125.17 88.47 3.7612 0.001324 0.006455-23.22 0.001480 0.004653-6.67 0.002293 0.002293 32.42 106.05 82.83 3.8498 79.23 72.56 3.9546 0.0 32.42 4.2008 0.0
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (6 reviews)
To calculate the density of gaseous CO2 at each of the given states using the perfect gas model we c...View the full answer

Answered By

Monette Taban
I am currently studying Computer Science Engineering, Due to my interest in programming languages and coding, I am interesetd on Technology so I search about it read about different types of technologies, I think my this habbis will help me to solve problems of students and that is why I am signing as a question answer expert.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Thermodynamics Fundamentals And Engineering Applications
ISBN: 9780521862738
1st Edition
Authors: William C. Reynolds, Piero Colonna
Question Posted:
Students also viewed these Engineering questions
-
In order to test the applicability of the ideal gas equation of state to calculate the density of saturated steam, compare the specific volume of saturated steam obtained from the steam table with...
-
(a) Calculate the density of the atmosphere at the surface of Mars (where the pressure is 650 Pa and the temperature is typically 253 K, with a CO 2 atmosphere), Venus (with an average temperature of...
-
Calculate the density of states g (E) for the free-electron model of a metal if E = 7.0eV and V = 1.0 cm3. Express your answer in units of states per electron volt.
-
Let be an arbitrary operation in Problems 5259. Describe the operation for each problem. 5038; 70 2= 9; 901 = 10; 8 0 2 = 10; -
-
If a potential investments internal rate of return is above the companys hurdle rate, should the investment be made?
-
The racism of eight White young adults, all convicted of committing an anti-Asian hate crime, was measured both before and after they had seen a film designed to reduce their racist attitudes. Using...
-
Suppose that \(v_{1}, v_{2}, \ldots, v_{n}\) are positive numbers. The arithmetic mean and the geometric mean of these numbers are, respectively, (a) It is always true that \(v_{A} \geq v_{G}\)....
-
Listed below are altitudes (thousands of feet) and outside air temperatures (degrees Fahrenheit) recorded by the author during Delta Flight 1053 from New Orleans to Atlanta. Is there sufficient...
-
JetBlue was able to drive up perceived customer value while lowering costs. This allowed it to carve out a strong strategic position and move to a non-contested market space. Then things went awry....
-
Given the following production plan, use a (a) Chase production strategy (b) Level production strategy to compute the monthly production, ending inventory / (backlog) and workforce levels. A worker...
-
Propane is in a 5 m 3 container as a saturated vapor at 300 K. The sealed tank is then cooled to 260 K by fresh snow. What are the pressure (Pa) and liquid volume in this 260 K state, and how much...
-
Consider each matrix as the augmented matrix of a linear system. State in words the next two elementary row operations that should be performed in the process of solving the system. 1-6 4 2 -7 0 0...
-
Why will Minnie not produce a quantity at which the market demand for water is inelastic?
-
What do you understand by quasi-static process? How it is achieved?
-
Distinguish between the term 'change of state', 'path', and 'process'.
-
Derive an expression for the first law of thermodynamics applied to a closed system. Define the internal energy of a system.
-
State the zeroth law of thermodynamics and first law of thermodynamics.
-
Define work. Show that work done \(W=P d V\).
-
Explain how technology facilitated the implementation of category management.
-
A regular deposit of $100 is made at the beginning of each year for 20 years. Simple interest is calculated at i% per year for the 20 years. At the end of the 20-year period, the total interest in...
-
A world-class runner can run half a mile in a time of 1 min and 45 s. What is the runners average speed in m/s?
-
One U.S. gallon is equivalent to 0.1337 ft 3 , 1 ft is equivalent to 0.3048 m, and 1000 L are equivalent to 1 m 3 . By using those definitions, determine the conversion factor between gallons and...
-
A passenger automobile is advertised as having a fuel economy rating of 29 mi/gal for highway driving. Express the rating in the units of km/L.
-
Select all that are not true Having televisions on where customers can see and hear them only serves to frustrate those in line Waiting in a longer line with a chance that the agent will allow you to...
-
While reviewing the data from the performance appraisal system, you find that managers generally provide the same rating for an employee across the three dimensions of job performance. That is, if...
-
a) what is the friction factor in pipe? b) what is the headloss in (m) due to friction in the pipe? Use Darcy - Weisbach Equation c) The liquid in the manometer has a specific gravity of 2.5....

Study smarter with the SolutionInn App


