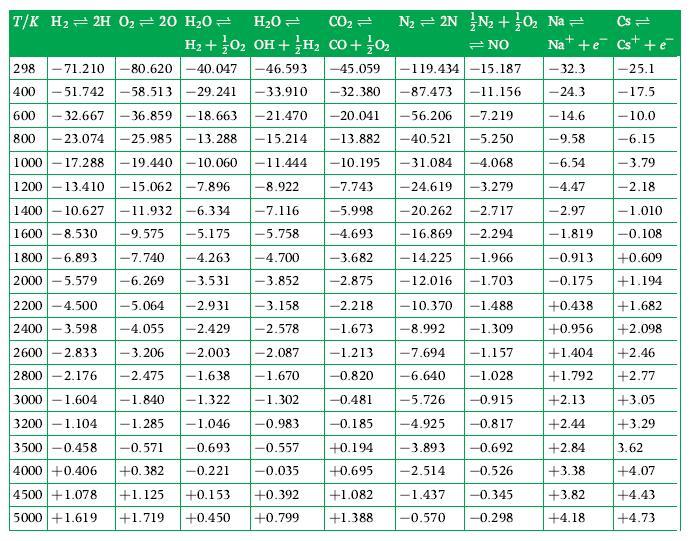
Using data for standardized enthalpy and absolute entropy obtained from GASMIX calculate the equilibrium constant for the
Question:
Using data for standardized enthalpy and absolute entropy obtained from GASMIX calculate the equilibrium constant for the gaseous reaction ![]() ?298 K. Derive an expression which would let you obtain this, as an alternative to using Table D.1 in terms of the equilibrium constants for simpler reactions.
?298 K. Derive an expression which would let you obtain this, as an alternative to using Table D.1 in terms of the equilibrium constants for simpler reactions.
Transcribed Image Text:
CO,+H, →→ CO+H,O
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 88% (9 reviews)
Given the chemical equation COH COHO and the equilibrium constant relation ...View the full answer

Answered By

Grace Igiamoh-Livingwater
I am a qualified statistics lecturer and researcher with an excellent interpersonal writing and communication skills. I have seven years tutoring and lecturing experience in statistics. I am an expert in the use of computer software tools and statistical packages like Microsoft Office Word, Advanced Excel, SQL, Power Point, SPSS, STATA and Epi-Info.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Thermodynamics Fundamentals And Engineering Applications
ISBN: 9780521862738
1st Edition
Authors: William C. Reynolds, Piero Colonna
Question Posted:
Students also viewed these Engineering questions
-
Impure nickel, refined by smelting sulfide ores in a blast furnace, can be converted into metal from 99.90% to 99.99% purity by the Mond process. The primary reaction involved in the Mond process is...
-
Calculate the equilibrium constant for the reaction O2 2O at temperatures of 298 K and 6000 K. Verify the result with Table A.11.
-
The phase diagram for SO2 is shown here. (a) What does this diagram tell you about the enthalpy change in the reaction SO2(I) SO2(g)? (b) Calculate the equilibrium constant for this reaction at 100...
-
A trader depreciates his vehicles at 40% per annum using the reducing balance method, with proportionate depreciation in the year of acquisition. In addition to vehicles acquired on 1 July 2010 for...
-
Two city councilors are debating whether to pursue a new project. Councilor Miles says it is only worth it to society if suppliers lower their costs to the city for the inputs to the project....
-
chidrens tickets are $20.00 adult tickets are $43.00 attendance was 1,100 profit was $35,800 how many chidren and how many adults
-
With reference to the previous exercise, construct a 95% confidence interval for the true mean inter-request time. Data From Previous Exercise 7.7 With reference to the n = 50 interrequest time ob-...
-
In the current year you are calculating a diversified companys deferred taxes. Based on an analysis of the companys current taxable income and pretax financial income, you have identified the...
-
Direction. Answer the question below. 1. Write the program code of the given flowchart. 2. Write the output of the program code. Activity 3. Writing program code Start Counter = 2 Counter
-
One way to see whether this procedure will be successful is to split the original data set into two subsets: one subset for estimation and one subset for validation. A regression equation is...
-
The burner of a power station must provide thermal power to the boiler at a rate of 88 MW. The maximum flame temperature is to be 1100 K, and the products of combustion can leave the boiler at no...
-
Find the mole fractions of O present in equilibrium O 2 at 1000 K and at 5000 K. Assume P = 1 atm. Repeat the calculation for N 2 and the same conditions.
-
What will the following program print when run? Select the one correct answer. (a) The program will print false true. (b) The program will print false false. (c) The program will print true false....
-
The common stock and debt of Northern Sludge are valued at $120 million and $80 million, respectively. Investors currently require a 13% return on the common stock and an 8% return on the debt....
-
Oriole Corporation purchased equipment very late in 2023. Based on generous capital cost allowance rates provided in the Income Tax Act, Oriole claimed CCA on its 2023 tax return but did not record...
-
Trade-Off vs. Pecking-Order Provide some of the strategies that companies use when they are in financial distress?
-
Calculate the following ratios for both 2021 and 2020 years: current ratio, quick ratio, debt to equity ratio, debt ratio, gross profit ratio, net profit ratio, return on equity, return on total...
-
As part of its stock-based compensation package, International Electronics granted 50 million stock appreciation rights (SARS) to top officers on January 1, 2024. At exercise, holders of the SARS are...
-
The S&P/TSX Composite Index rose 3.4%, dropped 1.4%, and then rose 2.1% in three successive months. The Index ended the three-month period at 9539. a. What was the Index at the beginning of the...
-
You are maintaining a subsidiary ledger account for Police-Training Expenditures for 2013. The following columns are used: Inventory purchases are initially recorded as expenditures. Record the...
-
How many milliliters of 53.4 (0.4) wt% NaOH with a density of 1.52 ( 0.01) g/mL will you need to prepare 2.000 L of 0.169 M NaOH? (b) If the uncertainty in delivering NaOH is [1] 0.01 mL, calculate...
-
(a) How many milliliters of 53.4 (0.4) wt% NaOH with a density of 1.52 ( 0.01) g/mL will you need to prepare 2.000 L of 0.169 M NaOH? (b) If the uncertainty in delivering NaOH is [1] 0.01 mL,...
-
We have a 37.0 ( 0.5) wt% HCl solution with a density of 1.18 ( 0.01) g/mL. To deliver 0.050 0 mol of HCl requires 4.18 mL of solution. If the uncertainty that can be tolerated in 0.050 0 mol is 2%,...
-
In the figure take q = 60 C, q2 = -35 C, and q = 19 C (Figure 1) Figure Part A y (m) 2- 093 91 92 T x (m) 2 3 1 of 1 > Find the electric force on q3. Enter your answers numerically separated by a...
-
The circular arc shown in the figure below has a uniform charge per unit length of 3.27 x 108 C/m. Find the potential at P, the center of the circle. (Take R 1.99 m.) 60.0 R P
-
You are a Hispanic student which English is your second language. At this point in your college career, I am sure that you have all written more papers than you care to count and you have definitely...

Study smarter with the SolutionInn App


